Market Share
Viral Vectors and Plasmid DNA Manufacturing Market Share Analysis
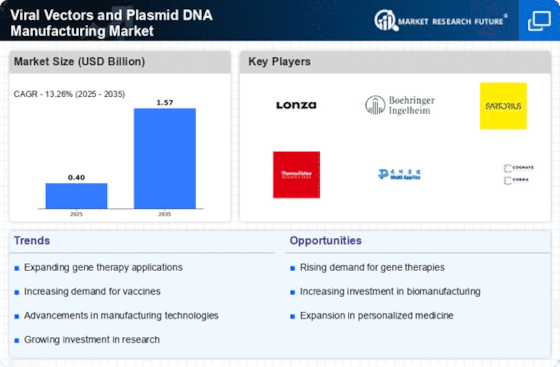
The viral vectors and plasmid DNA manufacturing market is an important segment inside the biopharmaceutical enterprise, serving as a basis for gene remedy and vaccine improvement. Companies in this area rent numerous strategies to establish and decorate their market proportion, recognizing the pivotal function of viral vectors and plasmid DNA in advancing scientific treatments. Adhering to Good Manufacturing Practices (GMP) is an essential method to ensure product satisfaction and regulatory compliance. Companies put money into sturdy, high-quality control structures, documentation, and centers to fulfill the stringent requirements of regulatory authorities. GMP compliance enhances market belief and allows broader market recognition. Recognizing the numerous healing applications of viral vectors and plasmid DNA and presenting custom-designed answers is a strategic approach. Tailoring products to therapeutic desires, whether for gene remedy or vaccine improvement, complements market relevance and guarantees that groups can deal with the particular requirements of their customers. Given the developing demand for viral vectors and plasmid DNA, ability enlargement and scalability are key techniques. Companies spend money on infrastructure and technology that allow for expanded production ability, ensuring they can meet the growing demands of the biopharmaceutical industry. Forming strategic partnerships and collaborations with biopharmaceutical organizations and studies institutions is a common approach. These collaborations can also include joint studies and development efforts, co-advertising and marketing agreements, or extraordinary delivery arrangements. Such partnerships now not only decorate market presence but also facilitate information alternatives and innovation. Expanding market presence globally is important for organizations in this quarter. Understanding nearby rules, adapting products to various healthcare systems, and setting up a strong distribution network make contributions to a hit global market penetration. A nicely carried out international growth approach guarantees that viral vectors and plasmid DNA merchandise are reachable on a worldwide scale. Ensuring regulatory compliance and preserving awesome requirements are non-negotiable for market positioning. Companies put money into acquiring vital certifications and adhering to stringent exceptional management measures to fulfill enterprise and regulatory necessities. Compliance no longer most effectively builds belief but also minimizes market access barriers. Educational initiatives and schooling applications for researchers and biopharmaceutical experts are crucial to market positioning. Companies that provide resources and education on the right use and handling of viral vectors and plasmid DNA foster an informed consumer base. This method now not only complements customer pride but also establishes long-term partnerships.



















Leave a Comment