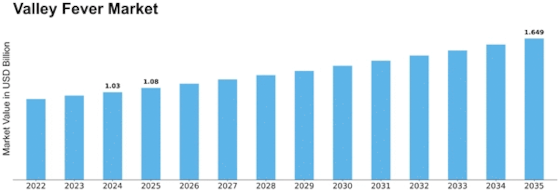
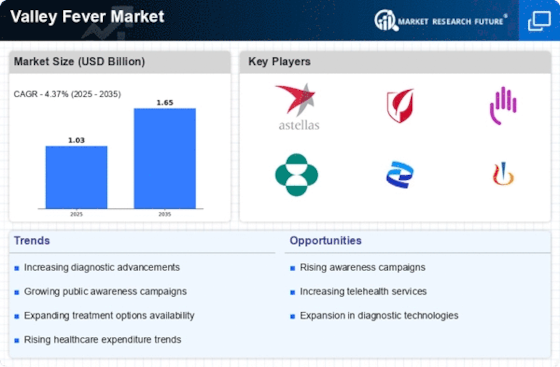
Valley Fever Size
Valley Fever Market Growth Projections and Opportunities
The occurrence of Valley Fever is emphatically affected by geographic elements. Districts with dry environments, like the southwestern US and portions of Mexico and Central America, experience higher paces of Valley Fever. The geographic pervasiveness shapes the interest for analytic devices and treatment choices here. The growth of populations in widespread areas adds to the general market elements. Expanding population density in regions inclined to Valley Fever might prompt a larger number of cases, driving the interest for successful antifungal prescriptions and symptomatic arrangements. Certain occupations, like development and horticulture, represent a higher risk of openness to Valley Fever. The word related risk factor impacts the interest for preventive measures, early finding, and focused medicines, molding the market for Valley Fever arrangements. The improvement of new antifungal medications is a vital consider the Valley Fever market. Progressing exploration and drug developments add to the extension of treatment choices, tending to difficulties like medication impediment and accompanying effects. The accessibility and openness of healthcare offices in widespread areas add to the market scene. Sufficient healthcare framework guarantees convenient determination and therapy, influencing the general interest for Valley Fever-related clinical benefits and items. Changes in environment examples can impact the spread and pervasiveness of Valley Fever. Modifications in temperature and precipitation levels might affect the environment of the parasite causing the illness, possibly prompting shifts in geographic dispersion, and influencing the market for Valley Fever arrangements. Surprising worldwide health emergencies, like pandemics, can disturb healthcare frameworks and effect the Valley Fever market. Changes in needs, asset distribution, and healthcare works on during emergencies might have suggestions for the analysis and treatment of Valley Fever.



















Leave a Comment