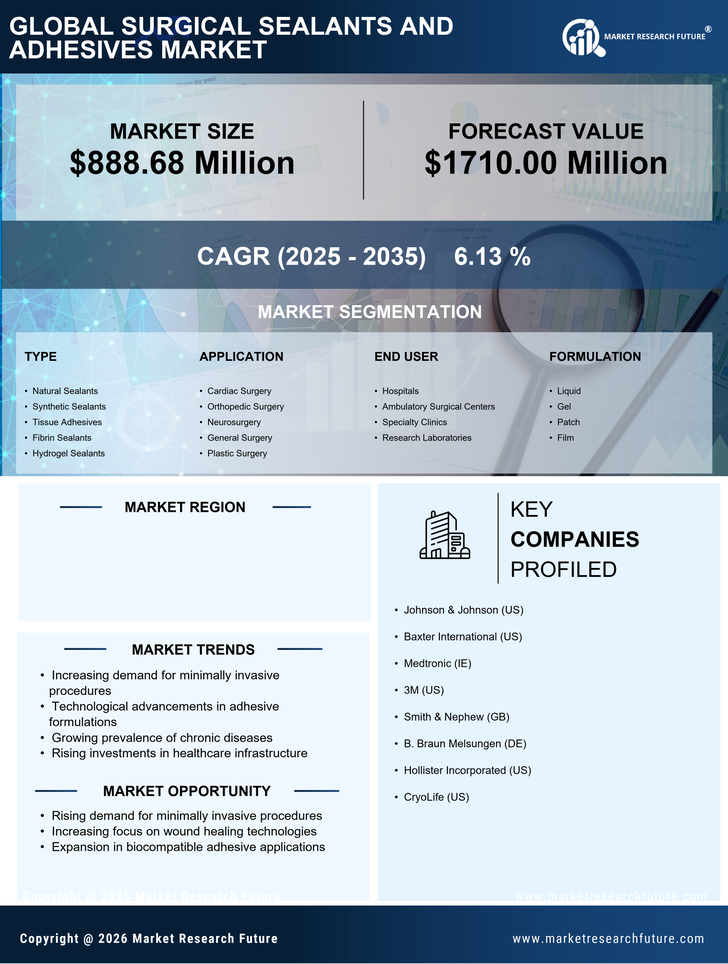
US Industrial Automation & Equipment Market Segmentation
Industrial Automation & Equipment By Type (USD Million, 2025-2035)
- Natural Sealants
- Synthetic Sealants
- Tissue Adhesives
- Fibrin Sealants
- Hydrogel Sealants
Industrial Automation & Equipment By Application (USD Million, 2025-2035)
- Cardiac Surgery
- Orthopedic Surgery
- Neurosurgery
- General Surgery
- Plastic Surgery
Industrial Automation & Equipment By End User (USD Million, 2025-2035)
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
- Research Laboratories
Industrial Automation & Equipment By Formulation (USD Million, 2025-2035)
- Liquid
- Gel
- Patch
- Film