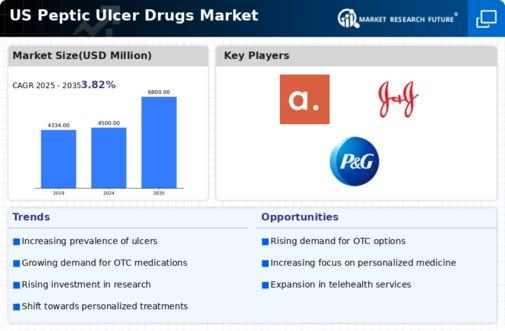
US Chemicals and Materials Market Segmentation
Chemicals and Materials By Pharmacological Class (USD Million, 2025-2035)
- Proton pump inhibitors (PPIs)
- Anti-histamines
- H2 antagonists
- Antacids
- Others
Chemicals and Materials By Clinical (USD Million, 2025-2035)
- Gastric ulcers
- Duodenal ulcers
- Esophageal ulcers