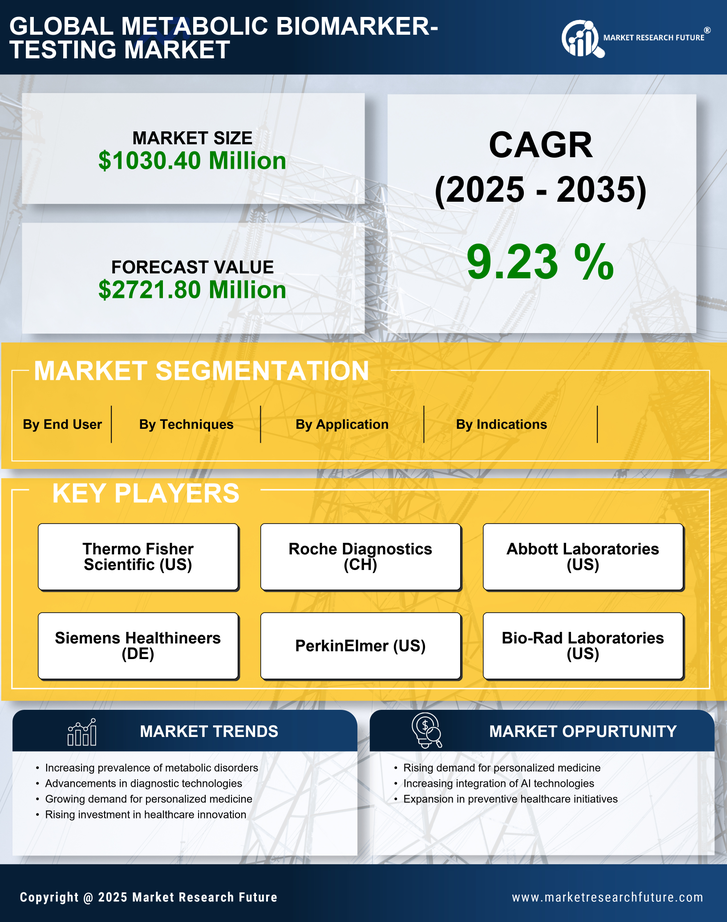
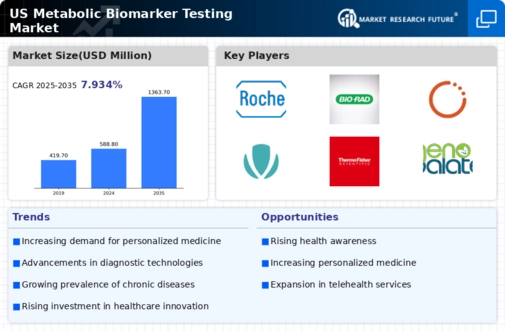
US Security, Access Control and Robotics Market Segmentation
Security, Access Control and Robotics By Techniques (USD Million, 2025-2035)
- Separation Techniques
- Detection Techniques
Security, Access Control and Robotics By Indications (USD Million, 2025-2035)
- Cancer
- Cardiovascular Disorders
- Neurological Disorders
- Inborn Errors of Metabolism
- Others
Security, Access Control and Robotics By Application (USD Million, 2025-2035)
- Drug Discovery or Drug Assessment
- Nutrigenomics
- Toxicology Testing
- Personalized Medicine
- Functional Genomics
- Others
Security, Access Control and Robotics By End User (USD Million, 2025-2035)
- Pharma & Biotech Companies
- Diagnostic Tool Companies
- Healthcare IT/Big Data Companies
- Clinical Laboratories
- Others