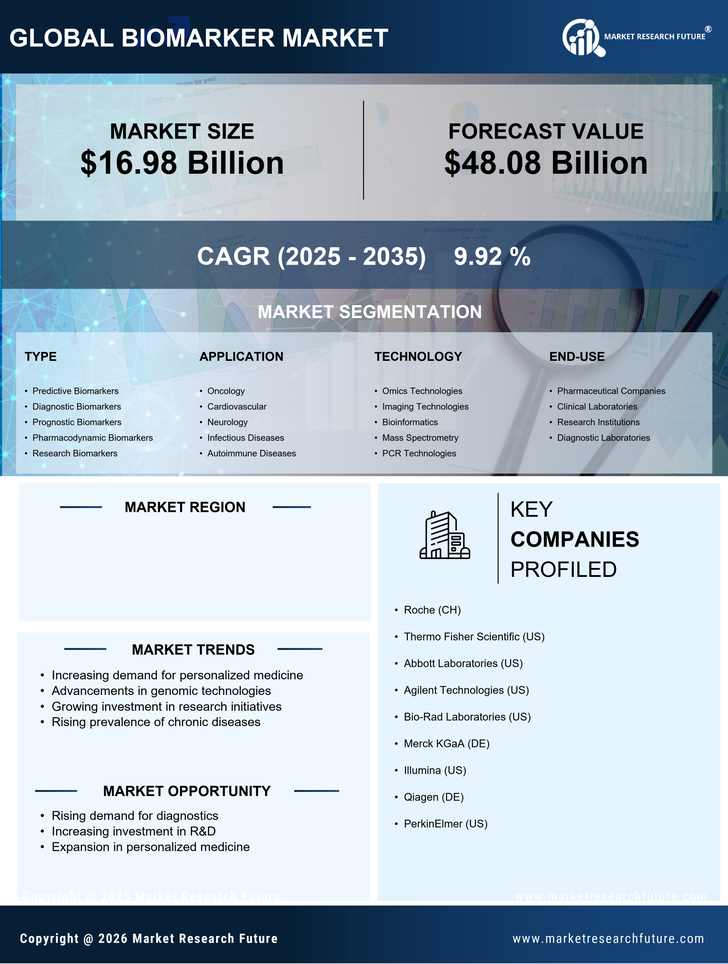
US Pharmaceutical Market Segmentation
Pharmaceutical By Type (USD Billion, 2025-2035)
- Predictive Biomarkers
- Diagnostic Biomarkers
- Prognostic Biomarkers
- Pharmacodynamic Biomarkers
- Research Biomarkers
Pharmaceutical By Application (USD Billion, 2025-2035)
- Oncology
- Cardiovascular
- Neurology
- Infectious Diseases
- Autoimmune Diseases
Pharmaceutical By Technology (USD Billion, 2025-2035)
- Omics Technologies
- Imaging Technologies
- Bioinformatics
- Mass Spectrometry
- PCR Technologies
Pharmaceutical By End-use (USD Billion, 2025-2035)
- Pharmaceutical Companies
- Clinical Laboratories
- Research Institutions
- Diagnostic Laboratories