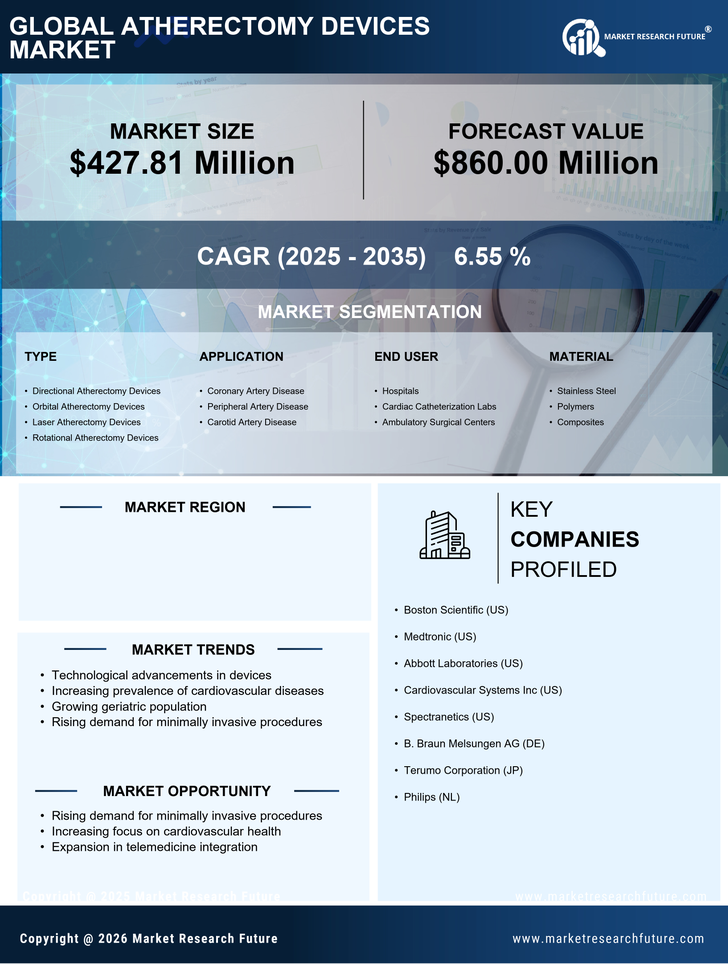
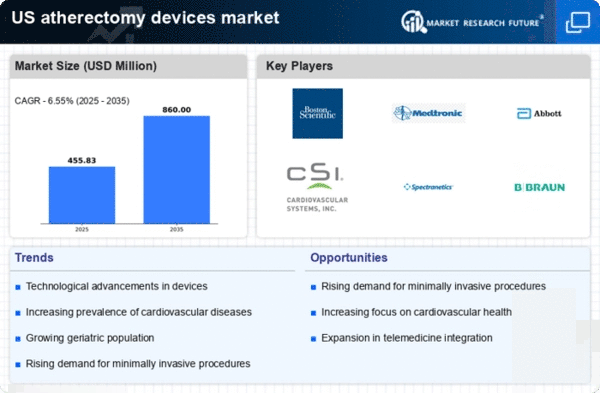
US Medical Device Market Segmentation
Medical Device By Type (USD Million, 2025-2035)
- Directional Atherectomy Devices
- Orbital Atherectomy Devices
- Laser Atherectomy Devices
- Rotational Atherectomy Devices
Medical Device By Application (USD Million, 2025-2035)
- Coronary Artery Disease
- Peripheral Artery Disease
- Carotid Artery Disease
Medical Device By End User (USD Million, 2025-2035)
- Hospitals
- Cardiac Catheterization Labs
- Ambulatory Surgical Centers
Medical Device By Material (USD Million, 2025-2035)
- Stainless Steel
- Polymers
- Composites