Urea Cycle Disorder Market
ID: MRFR/HC/40527-HCR
100 Pages
Rahul Gotadki
Last Updated: April 06, 2026
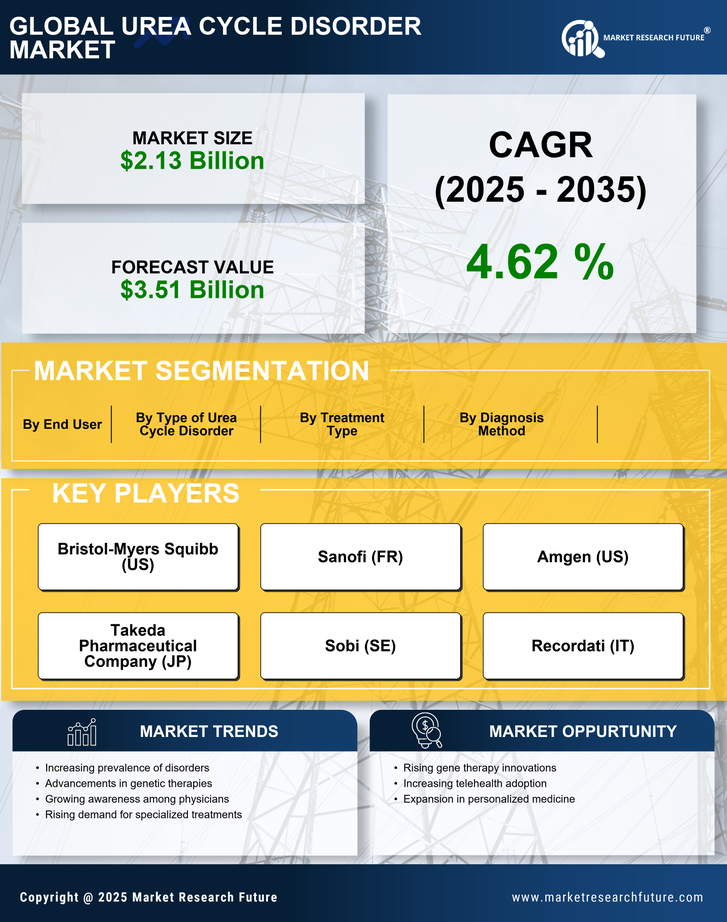
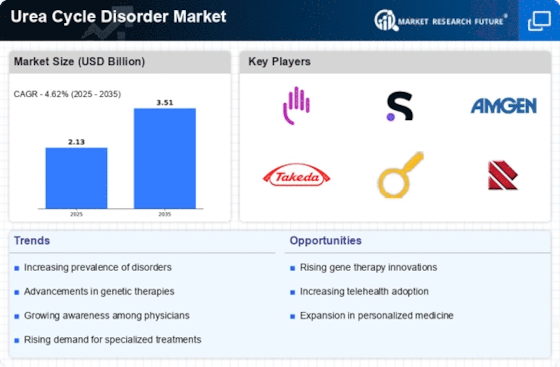
Urea Cycle Disorder Market Research Report By Type of Urea Cycle Disorder (Citrullinemia, Argininosuccinic Aciduria, Ornithine Transcarbamylase Deficiency, Hyperammonaemia, Carbamoyl Phosphate Synthetase Deficiency), By Diagnosis Method (Blood Tests, Urine Tests, Genetic Testing, Newborn Screening, Imaging Techniques), By Treatment Type (Dietary Management, Medications, Liver Transplant, Gene Therapy, Supportive Care), By End User (Hospitals, Specialized Clinics, Home Care Settings, Research Institutions) and By Regional (North America, Europe, South America, Asia Pacific, Middle East and Africa) - Growth & Industry Forecast 2025 To 2035