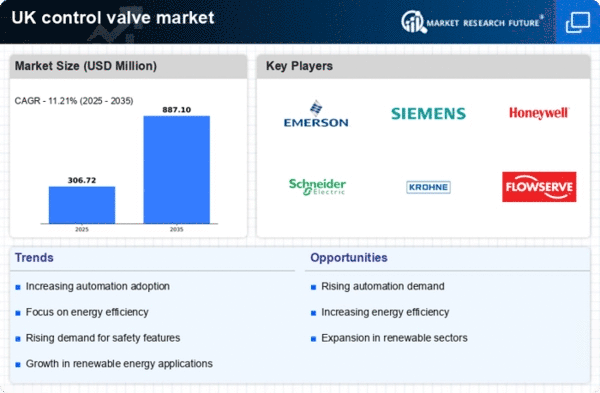
UK Medical Device Market Segmentation
Medical Device By Valve Type (USD Million, 2025-2035)
- Rotary valves
- Ball valves
- Butterfly valves
- Plug valve
- Linear valves
- Globe valves
- Diaphragm valves
- Others
Medical Device By Material (USD Million, 2025-2035)
- Stainless Steel
- Cast Iron
- Alloy-based
- Cryogenic
- Others
Medical Device By Valve Size (USD Million, 2025-2035)
- BELOW 1”
- 1” TO 6”
- 25” TO 50”
- ABOVE 50”
Medical Device By Component (USD Million, 2025-2035)
- Valve Body
- Actuator
- Others
Medical Device By End-Use (USD Million, 2025-2035)
- Oil & Gas
- Water & Wastewater Treatment
- Energy & Power
- Chemicals
- Food & Beverage
- Others
Medical Device By Technology (USD Million, 2025-2035)
- Hall effect sensor
- Digital Fieldbus protocols
Medical Device By Vertical (USD Million, 2025-2035)
- Pharma
- Food and Beverage
- Chemicals
- Energy and Power
- Water and Wastewater Treatment
- Oil and Gas