Market Trends
Key Emerging Trends in the Transcatheter Embolization and Occlusion Devices Market
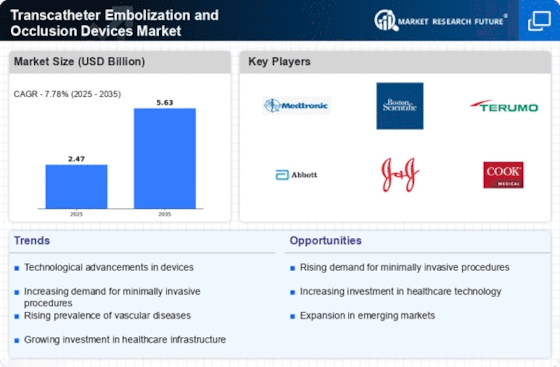
The Transcatheter Embolisation and Occlusion Devices Market reflects substantial trends associated with evolving interventional radiology techniques; growing incidences of vascular problems; as well as an upsurge in preference for minimally invasive procedures. One significant trend is the rising adoption of transcatheter embolisation and occlusion devices for treatment of different vascular conditions. These include embolic coils, liquid embolics, or vascular plugs that can block or close blood vessels via minimally invasive catheter-based procedures. This transition towards transcatheter approaches arises from advantages like less invasiveness, shorter recovery timeframes and fewer complications compared to traditional surgery.
Market dynamics are shaped by technological advances in embolization & occlusion devices. Manufacturers are concentrating on developing gadgets that have better accuracy, deliverability and compatibility with diverse anatomical structures. Additionally the safety & effectiveness of these devices has been increased through use of novel materials such as detachable coils or bioresorbable embolics Among other things imaging technologies such cone-beam computed tomography (CBCT) helps during transcatheter procedures by improving visualization leading to accurate device placement.
Transcatheter embolic agents including occlusive devices are increasingly employed in the treatment of vascular malformations, aneurysms and tumors. As the diseases become more prevalent, transvenous approaches provide targeted solutions to them. Embolic agents together with occlusion devices are essential tools in handling conditions such as uterine fibroids, arteriovenous malformations (AVMs) and hepatic tumors thereby offering alternatives to surgery while reducing overall morbidity associated with these ailments.
The market is becoming diversified through an increasing range of embolic agents and occlusion devices. Manufacturers are producing a variety of devices for use in particular clinical scenarios, which enables healthcare providers to choose the best option that matches patient characteristics and nature of the disease. This customization trend towards devices increases flexibility during transcatheter procedures and therefore gives personalized touch towards patients’ healing care.
An increased application of transcatheter embolization and occlusion therapies is being witnessed by the market. In some instances physicians may combine embolic agents as well as occlusive devices in bid to achieve optimum results for complex vascular cases. These developments indicate changing treatment strategies in interventional radiology where multimodal therapy might be used so as to handle unique challenges posed by specific vascular conditions.

















Leave a Comment