Therapeutic Proteins Oral Vaccines Market
ID: MRFR/Pharma/26212-HCR
100 Pages
Rahul Gotadki
Last Updated: April 24, 2026
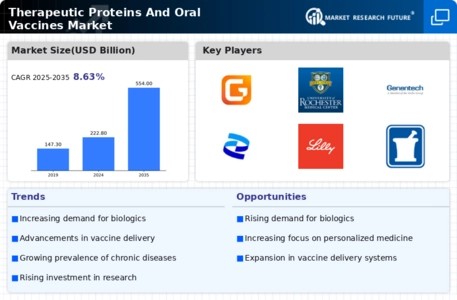
Therapeutic Proteins And Oral Vaccines Market Research Report By Therapeutic Class (Antibodies, Enzymes, Hormones, Growth factors, Polypeptides), By Vaccine Type (Live attenuated, Inactivated, Toxoid, Subunit, Conjugate), By Disease Indication (Cancer, Cardiovascular diseases, Autoimmune diseases, Infectious diseases, Neurological disorders), By Route of Administration (Intravenous, Subcutaneous, Intramuscular, Oral, Topical) and By Regional (North America, Europe, South America, Asia Pacific, Middle East and Africa) - Growth & Industry Forecast 2025 To 2035
.png)