Therapeutic Medical Guide Wire Size
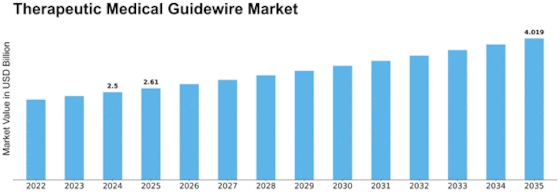
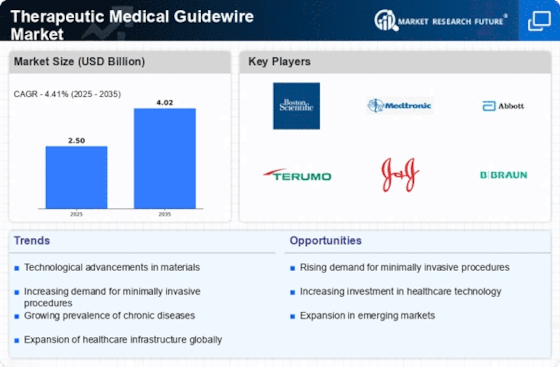
Therapeutic Medical Guide Wire Market Growth Projections and Opportunities
The Therapeutic Medical Guidewire market is shaped by various market factors that collectively influence its growth and dynamics. One of the key drivers propelling this market is the increasing prevalence of cardiovascular diseases. As the global burden of heart-related conditions rises, the demand for minimally invasive procedures, where therapeutic medical guidewires play a crucial role, is escalating. These guidewires assist healthcare professionals in navigating through blood vessels with precision during procedures like angioplasty and stent placement, contributing significantly to the market's expansion.
Technological advancements in medical devices are also instrumental in driving the therapeutic medical guidewire market. Ongoing innovations in guidewire design and materials lead to improved flexibility, maneuverability, and trackability, allowing for more intricate and precise procedures. The integration of advanced technologies enhances the overall efficacy of therapeutic interventions, attracting healthcare providers to adopt these cutting-edge guidewires in their practices.
Regulatory considerations are another critical factor influencing the therapeutic medical guidewire market. Stringent regulations ensure the safety and efficacy of medical devices, including guidewires, and compliance with these standards is essential for market entry and sustained growth. Changes in regulatory requirements, such as updated quality standards or approvals for new indications, can impact the market landscape by influencing product development timelines and market access.
Economic factors also play a role in shaping the market dynamics. The affordability and accessibility of therapeutic medical guidewires are influenced by economic conditions and healthcare spending. Economic downturns may impact hospitals' budgets and purchasing decisions, affecting the adoption rate of advanced medical devices like guidewires. Conversely, economic growth and increased healthcare expenditure may drive investment in state-of-the-art medical technologies, positively impacting the market.
Furthermore, the market is influenced by the evolving landscape of healthcare delivery. A growing preference for minimally invasive procedures over traditional surgeries contributes to the demand for therapeutic medical guidewires. These guidewires enable healthcare professionals to perform complex procedures with smaller incisions, leading to reduced recovery times and improved patient outcomes. As healthcare systems continue to prioritize less invasive approaches, the market for therapeutic medical guidewires is expected to witness sustained growth.
Competition within the market is another notable factor. The presence of multiple manufacturers and suppliers fosters innovation and encourages the development of guidewires with enhanced features and functionalities. Competitive forces can lead to improvements in product quality, pricing strategies, and customer service, ultimately benefiting healthcare providers and patients seeking advanced therapeutic solutions.
Market factors are also influenced by healthcare professionals' awareness and training. As clinicians become more familiar with the advantages of using therapeutic medical guidewires in various medical procedures, the demand for these devices is likely to increase. Educational initiatives by manufacturers and healthcare institutions can contribute to the broader understanding of guidewire applications, driving market growth.


















Leave a Comment