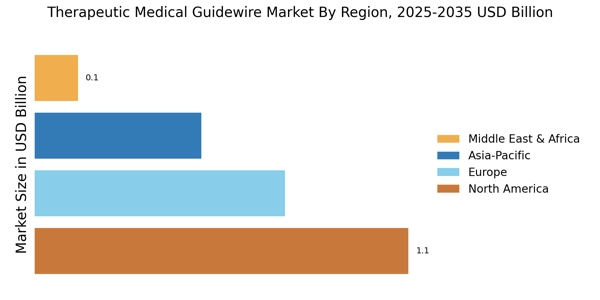
Increasing Prevalence of Chronic Diseases
The rising prevalence of chronic diseases such as cardiovascular disorders, diabetes, and cancer is a significant driver for the Therapeutic Medical Guidewire Market. As these conditions necessitate various interventional procedures, the demand for effective guidewires is expected to escalate. According to recent statistics, cardiovascular diseases alone account for nearly 31% of all global deaths, underscoring the urgent need for advanced therapeutic solutions. This growing patient population is likely to propel the market forward, as healthcare providers seek to adopt innovative guidewire technologies to improve patient outcomes. Consequently, the market is anticipated to expand as healthcare systems adapt to the increasing burden of chronic diseases.
Technological Innovations in Guidewire Design
The Therapeutic Medical Guidewire Market is experiencing a surge in technological innovations that enhance the performance and efficacy of guidewires. Recent advancements include the development of hydrophilic coatings and advanced materials that improve flexibility and maneuverability. These innovations are crucial for navigating complex anatomical structures, thereby increasing the success rates of various therapeutic procedures. The introduction of smart guidewires equipped with sensors for real-time feedback is also gaining traction. Such technologies not only improve procedural outcomes but also reduce the risk of complications. As a result, the market is projected to witness a compound annual growth rate of approximately 6.5% over the next few years, driven by these technological advancements.
Rising Adoption of Minimally Invasive Techniques
The shift towards minimally invasive surgical techniques is profoundly influencing the Therapeutic Medical Guidewire Market. These techniques offer numerous advantages, including reduced recovery times, lower risk of infection, and minimal scarring. As healthcare providers increasingly recognize the benefits of such approaches, the demand for specialized guidewires designed for minimally invasive procedures is on the rise. Market data indicates that minimally invasive surgeries are projected to grow at a rate of 7% annually, further driving the need for advanced guidewire solutions. This trend not only enhances patient satisfaction but also aligns with the broader healthcare objective of improving surgical outcomes while minimizing patient trauma.
Regulatory Support for Medical Device Innovations
Regulatory support for medical device innovations is playing a crucial role in shaping the Therapeutic Medical Guidewire Market. Regulatory bodies are increasingly streamlining the approval processes for new medical technologies, thereby encouraging manufacturers to invest in research and development. This supportive environment fosters innovation, allowing for the introduction of advanced guidewire designs that meet the evolving needs of healthcare providers. Furthermore, initiatives aimed at enhancing patient safety and efficacy are likely to drive the adoption of new guidewires in clinical settings. As regulatory frameworks continue to evolve, the market is expected to witness a surge in the introduction of novel guidewire technologies, ultimately benefiting patient care.
Expansion of Healthcare Infrastructure in Developing Regions
The expansion of healthcare infrastructure in developing regions is emerging as a pivotal driver for the Therapeutic Medical Guidewire Market. As countries invest in improving their healthcare systems, the availability of advanced medical technologies, including therapeutic guidewires, is increasing. This growth is particularly evident in regions where access to quality healthcare has been historically limited. The establishment of new hospitals and clinics is likely to facilitate the adoption of innovative medical devices, including guidewires, thereby enhancing treatment options for patients. As a result, the market is expected to benefit from this infrastructural development, with a projected increase in demand for guidewires in these emerging markets.