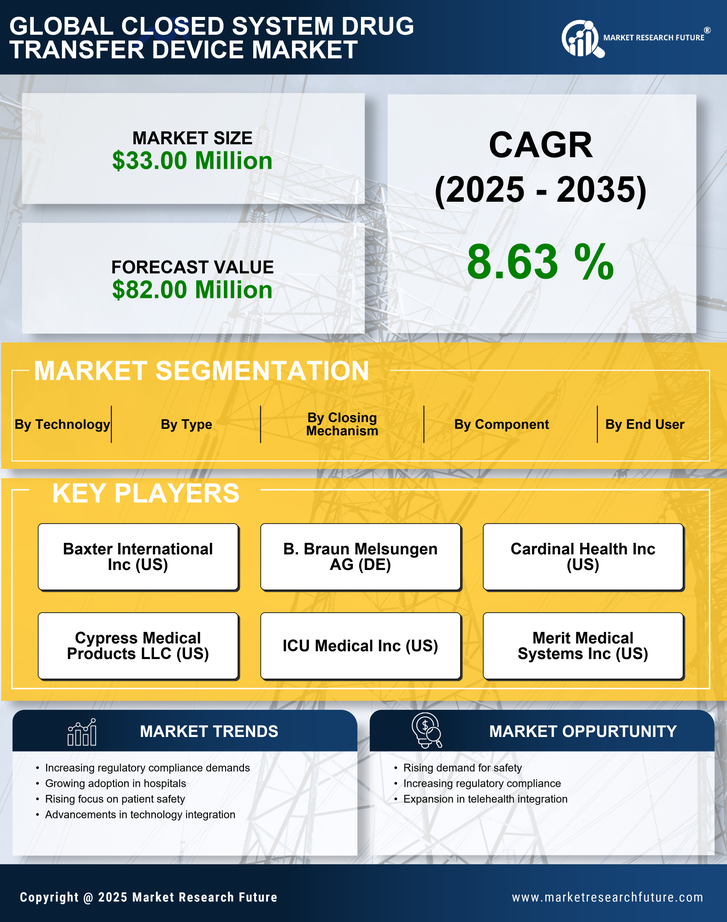
Spain Medical Device Market Segmentation
Medical Device By Type (USD Million, 2025-2035)
- Membrane-To-Membrane Systems
- Needleless Systems
Medical Device By Component (USD Million, 2025-2035)
- Vial Access Devices
- Syringe Safety Devices
- Bag/Line Access Devices
- Accessories
Medical Device By Closing Mechanism (USD Million, 2025-2035)
- Push To Turn System
- Color-To-Color Alignment System
- Luer Lock Systems
- Click-To Lock System
Medical Device By Technology (USD Million, 2025-2035)
- Compartmentalize Devices
- Diaphragm-Based Devices
- Air Filtration Devices
Medical Device By End User (USD Million, 2025-2035)
- Hospitals & clinics
- Specialty centers
- Others