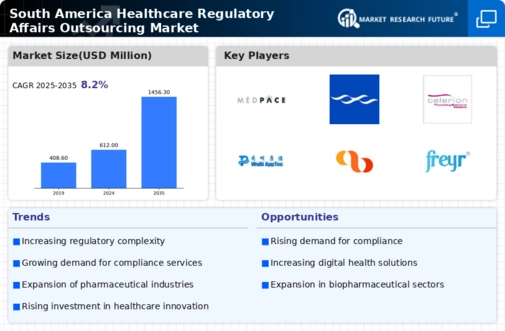
South America Healthcare Market Segmentation
Healthcare By Service Type (USD Billion, 2022-2035)
- Regulatory Consulting
- Regulatory Affairs Outsourcing
- Clinical Trial Application Management
- Post-Market Surveillance
- Product Registration
Healthcare By Therapeutic Area (USD Billion, 2022-2035)
- Pharmaceuticals
- Medical Devices
- Biotechnology
- Cosmetics
- Nutraceuticals
Healthcare By Client Type (USD Billion, 2022-2035)
- Pharmaceutical Companies
- Biotechnology Firms
- Medical Device Manufacturers
- Contract Research Organizations
- Academic Institutions
Healthcare By Regulatory Framework (USD Billion, 2022-2035)
- Local Regulations
- International Guidelines
- Compliance Management
- Quality Assurance
- Risk Assessment