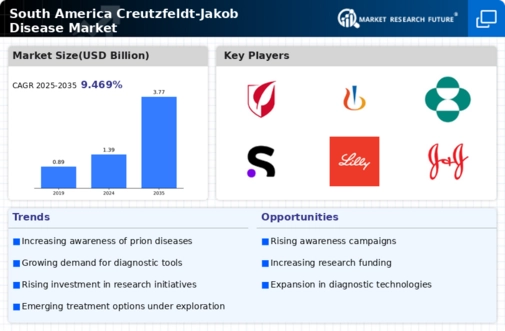
South America Medical Device Market Segmentation
Medical Device By Type (USD Million, 2025-2035)
- Sporadic Creutzfeldt-Jakob Disease
- Familial Creutzfeldt-Jakob Disease
- Iatrogenic Creutzfeldt-Jakob Disease
- Variant Creutzfeldt-Jakob Disease
Medical Device By Diagnosis Method (USD Million, 2025-2035)
- Electroencephalogram
- Magnetic Resonance Imaging
- Cerebrospinal Fluid Tests
- Brain Biopsy
Medical Device By Treatment Approach (USD Million, 2025-2035)
- Supportive Care
- Experimental Therapies
- Symptomatic Treatment
Medical Device By End User (USD Million, 2025-2035)
- Hospitals
- Diagnostic Laboratories
- Research Institutes