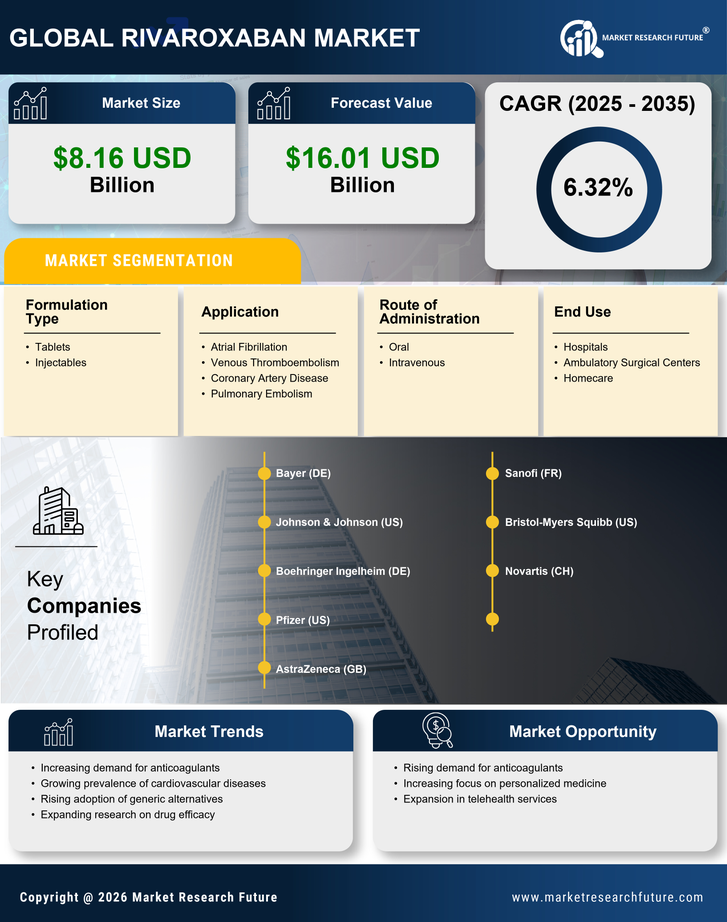
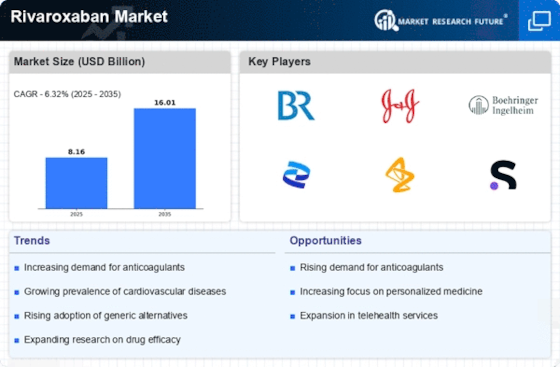
Healthcare Market Segmentation
Healthcare By Application (USD Billion, 2025-2035)
- Atrial Fibrillation
- Venous Thromboembolism
- Coronary Artery Disease
- Pulmonary Embolism
Healthcare By Route of Administration (USD Billion, 2025-2035)
- Oral
- Intravenous
Healthcare By Formulation Type (USD Billion, 2025-2035)
- Tablets
- Injectables
Healthcare By End Use (USD Billion, 2025-2035)
- Hospitals
- Ambulatory Surgical Centers
- Homecare