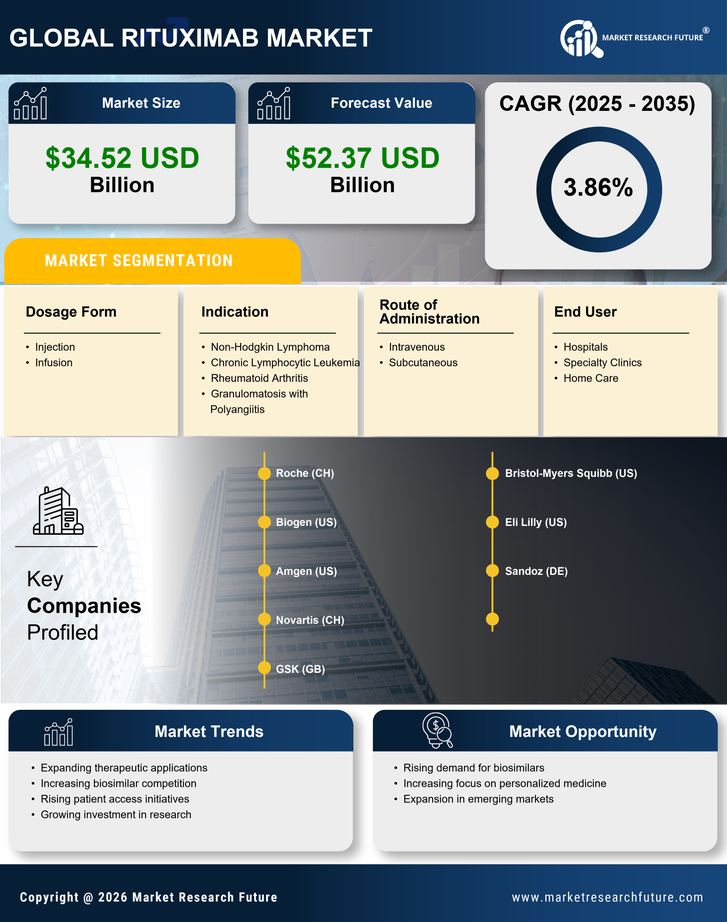
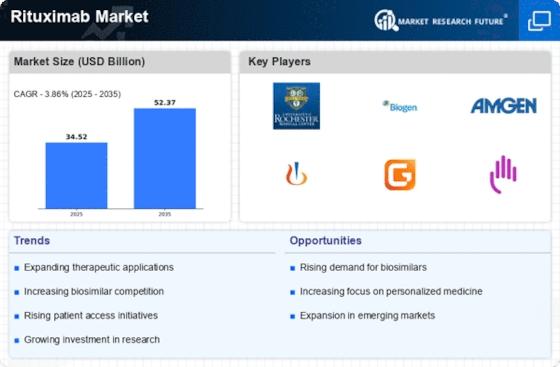
Healthcare Market Segmentation
Healthcare By Indication (USD Billion, 2025-2035)
- Non-Hodgkin Lymphoma
- Chronic Lymphocytic Leukemia
- Rheumatoid Arthritis
- Granulomatosis with Polyangiitis
Healthcare By Route of Administration (USD Billion, 2025-2035)
- Intravenous
- Subcutaneous
Healthcare By Dosage Form (USD Billion, 2025-2035)
- Injection
- Infusion
Healthcare By End User (USD Billion, 2025-2035)
- Hospitals
- Specialty Clinics
- Home Care