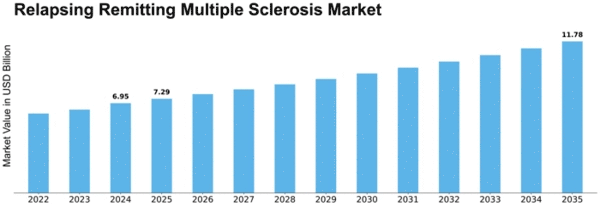
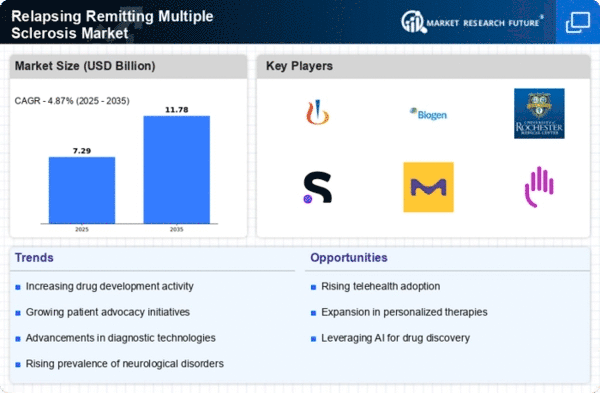
Relapsing Remitting Multiple Sclerosis Size
Relapsing Remitting Multiple Sclerosis Market Growth Projections and Opportunities
A plethora of market factors influence the progress and dynamics of Relapsing-Remitting Multiple Sclerosis (RRMS) Market. A major factor among them is the rapid increase in the prevalence and knowledge about multiple sclerosis as a chronic autoimmune disease. With improvement in diagnostic abilities and heightening of public awareness campaigns, more individuals are being diagnosed with RRMS due to which there is an increasing demand for effective treatment methods. This rise in awareness contributes significantly to the growth of RRMS treatment market.
Medical research advancements and pharmaceutical innovations are crucial driving factors in the RRMS market. On-going efforts by researchers plus pharmaceutical corporations have led to invention of advanced disease-modifying treatments that specifically target complex aspects posed by RRMS. Apart from enhancing efficacy levels on existing therapies, these improvements bring new therapeutic modalities thereby expanding the options that healthcare providers have for treating this condition. The dynamic nature of neurology medicine plays a significant role in propelling expansionary moves within RRMS treatment market.
Demographic factors such as MS prevalence rates and demographics of affected population greatly impact on RRMS-market dynamics.Knownly, women tend to be more frequently affected than men while majority MS cases are diagnosed between 20–50 years. These demographic factors will then influence markets through age or gender specific tailored treatments that address different patient groups’ concerns.
The availability as well as accessibility of healthcare infrastructure plus services shape its landscape.Section B Regions where neurocare facilities are strongly developed can ably diagnose and treat RRMS accordingly.On contrary weak care support systems may give rise to difficulties with timely provision of comprehensive care resulting in slowed market growth within such areas.
Reimbursement policies together with healthcare expenditure by governments and insurance companies play a critical role in influencing uptake patterns for RRMs treatments.A good reimbursement policy will encourage patients seek early medical intervention hence adhering to DMTs, usually expensive.On contrast inadequate reimbursement or limited coverage would limit access to some treatment options thereby impacting on the overall demand for such services.The economic burden of managing chronic conditions like RRMS further underscores the importance of effective reimbursement strategies.
Patient awareness and education are key elements that drive RRMS treatment market. Increasing awareness about MS symptoms, benefits of early intervention and availability of disease modifying therapies can result in earlier diagnosis and better patient outcomes. Besides these lifestyle modification efforts by healthcare providers who educate patients as well as promote adherence to treatment plans play a vital role in improving overalls condition management hence influencing markets positively.



















Leave a Comment