North America : Innovation and Market Leadership
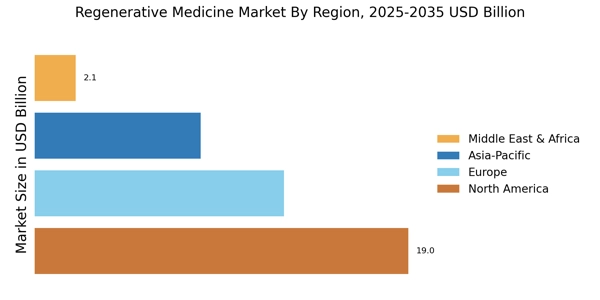
North America leads in the Regenerative Medicine Market Size, accounting for over 45.10% of the global revenue in 2024. Key growth drivers include advanced healthcare infrastructure, significant R&D investments, and a favorable regulatory environment. The U.S. leads this region, followed closely by Canada, which contributes around 10% to the market. Regulatory catalysts, such as expedited approval processes for regenerative therapies, further enhance market growth.
- CDC reports that chronic diseases account for 6 out of 10 deaths annually in the U.S., while over 130 million adults live with at least one chronic condition. High healthcare spending exceeding USD 4 trillion annually further supports rapid adoption of advanced regenerative therapies.
The competitive landscape is robust, with major players like Acelity, Organogenesis, and Vericel leading the charge. The presence of these regenerative medicine companies fosters innovation and collaboration, driving advancements in cell therapy and tissue engineering. The U.S. is home to numerous clinical trials and research initiatives, solidifying its position as a hub for regenerative medicine development. Overall, the North American market is characterized by a strong focus on innovation and patient-centric solutions.
Europe : Emerging Market with Potential
Europe Regenerative Medicine Market was valued at USD 12.639 Billion in 2024, making it the second-largest regional market with a 30% share. The region benefits from a strong emphasis on research and development, supported by government initiatives and funding. Countries like Germany and the UK are at the forefront, with Germany holding about 12% of the market. Regulatory frameworks, such as the Advanced Therapy Medicinal Products (ATMP) regulation, are pivotal in facilitating market expansion.
- ECDC reports chronic diseases contribute to nearly 80% of deaths in Europe, while WHO estimates over 3.7 million cancer cases annually in the region. Increasing public healthcare investments and innovation funding are accelerating development and accessibility of regenerative therapies.
Leading countries in Europe are investing heavily in regenerative medicine, with key players like Tissue Regenix and Mesoblast making notable contributions. The competitive landscape is evolving, with a mix of established companies and innovative startups. Collaborative efforts between academia and industry are fostering advancements in cell and gene therapies, positioning Europe as a significant player in the global regenerative medicine arena. The region's commitment to innovation and regulatory support is expected to drive future growth.
Asia-Pacific : Rapid Growth and Investment
Asia-Pacific is rapidly emerging as a significant player in the regenerative medicine market, holding approximately 20% of the global share. The region is driven by increasing healthcare expenditures, a growing aging population, and rising demand for advanced medical treatments. Countries like Japan and Australia are leading the charge, with Japan contributing around 8% to the market. Regulatory reforms aimed at accelerating the approval of regenerative therapies are also boosting market dynamics.
The competitive landscape in Asia-Pacific is characterized by a mix of local and international players, with companies like Mesoblast and Sernova making strides in the market. The region is witnessing a surge in clinical trials and research initiatives, supported by government funding and partnerships. As the demand for innovative therapies continues to rise, Asia-Pacific is poised for substantial growth in the regenerative medicine sector, driven by technological advancements and increased investment in healthcare infrastructure.
Middle East and Africa : Untapped Potential and Growth
The Middle East and Africa region is gradually emerging in the regenerative medicine market, currently holding about 5% of the global share. Key growth drivers include increasing healthcare investments, a rising prevalence of chronic diseases, and a growing interest in advanced medical technologies. Countries like South Africa and the UAE are leading the market, with the UAE focusing on becoming a hub for medical innovation. Regulatory frameworks are evolving to support the introduction of regenerative therapies.
The competitive landscape is still developing, with a mix of local startups and international companies exploring opportunities in the region. The presence of key players is limited, but there is a growing interest in partnerships and collaborations to enhance research and development efforts. As healthcare systems in the region continue to evolve, the potential for growth in regenerative medicine is significant, driven by increasing awareness and investment in regenerative medical solutions.