Point of Care Diagnostics Testing Market Summary
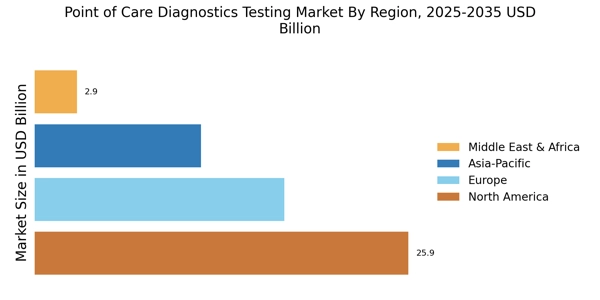
According to Market Research Future analysis, the Point of Care Diagnostics Testing Market Size was valued at USD 57.71 Billion in 2024 & the market is projected to grow from USD 63.24 Billion in 2025 to USD 157.87 Billion by 2035, registering a CAGR of 9.58% during the forecast period (2025–2035). North America led the market with over 44.88% share, generating around USD 25.9 billion in revenue.
Rising demand for rapid, accessible diagnostic solutions and increasing prevalence of chronic and infectious diseases are key growth drivers of the point of care diagnostics testing market. Advancements in portable technologies and decentralized healthcare delivery are accelerating global adoption significantly.
According to WHO, over 1 billion people require diagnostic testing annually for infectious diseases, while CDC reports that 6 in 10 adults in the U.S. have at least one chronic condition, driving strong demand for rapid point-of-care diagnostic solutions globally.
Key Market Trends & Highlights
The Point of Care Diagnostics Testing Market is experiencing robust growth driven by technological advancements and a shift towards decentralized healthcare.
- North America accounts for over 44.88% market share in 2024, driven by high chronic disease prevalence.
- Europe market valued at USD 17.31 billion in 2024, holding 30% share with strong preventive healthcare initiatives.
- Over 1 billion diagnostic tests conducted annually globally, significantly driving demand for rapid point-of-care diagnostic solutions.
- Glucose monitoring dominates with 38% share due to rising diabetes prevalence and need for continuous monitoring solutions.
Market Size & Forecast
| 2024 Market Size | 57.71 (USD Billion) |
| 2035 Market Size | 157.87 (USD Billion) |
| CAGR (2025 - 2035) | 9.58% |
Major Players
Companies such as Abbott Laboratories (US), Roche Diagnostics (CH), Siemens Healthineers (DE), Danaher Corporation (US), Thermo Fisher Scientific (US), Becton Dickinson and Company (US), Cepheid (US), Quidel Corporation (US), Hologic, Inc. (US) are some of the major participants in the global market.