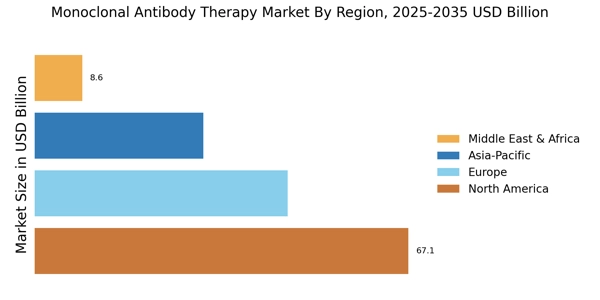
The Monoclonal Antibody Therapy Market is currently experiencing a transformative phase, characterized by rapid advancements in biotechnology and an increasing understanding of disease mechanisms. This evolution is largely driven by the growing prevalence of chronic diseases and the rising demand for targeted therapies.
As healthcare systems worldwide strive to enhance patient outcomes, monoclonal antibodies are emerging as a pivotal component in the treatment landscape, offering specificity and efficacy that traditional therapies may lack. Furthermore, the ongoing research and development efforts are likely to expand the therapeutic applications of these agents, potentially addressing a broader range of conditions than previously thought.
In addition to the scientific advancements, the Monoclonal Antibody Therapy Market is also influenced by regulatory changes and the increasing collaboration between top monoclonal antibody companies and research institutions. These partnerships appear to foster innovation and expedite the development of new therapies.
Moreover, the market is witnessing a shift towards personalized medicine, where treatments are tailored to individual patient profiles, enhancing the overall effectiveness of therapies. As the landscape continues to evolve, stakeholders must remain vigilant to adapt to emerging trends and challenges, ensuring that they can capitalize on the opportunities presented by this dynamic market.
Rise of Personalized Medicine
The Monoclonal Antibody Therapy Market is increasingly leaning towards personalized medicine, where treatments are customized based on individual patient characteristics. This trend suggests a shift from one-size-fits-all approaches to more tailored therapies, potentially improving treatment outcomes and minimizing adverse effects.
Growing precision medicine adoption is accelerating monoclonal antibody demand. World Health Organization notes hundreds of monoclonal antibodies currently under clinical development targeting oncology, immunology, and infectious diseases, highlighting strong pipeline expansion and increasing patient-specific therapeutic solutions across global healthcare systems.
Technological Advancements in Production
The antibody market analysis suggests that innovations in biomanufacturing technologies are reshaping the Monoclonal Antibody Therapy Market. Enhanced production methods may lead to more efficient and cost-effective manufacturing processes, thereby increasing the availability of these therapies and reducing overall healthcare costs.
Advancements in monoclonal antibody development are accelerating approvals. In 2024, 50 novel drugs were approved globally, including 16 biologics and 13 monoclonal antibodies, representing the highest approval rate since 2015, reflecting strong technological innovation and expanding therapeutic pipeline.
Expansion into Emerging Markets
The Monoclonal Antibody Therapy Market is witnessing a notable expansion into emerging markets. This trend indicates a growing recognition of the potential for monoclonal antibodies in regions with increasing healthcare investments, thereby broadening access to advanced therapies for diverse populations, which augments the antibody market size.
Emerging markets are expanding monoclonal antibody adoption due to increasing disease burden. Institute for Health Metrics and Evaluation indicates chronic diseases account for nearly 74% of global deaths, driving healthcare investments and expanding access to biologics across developing healthcare systems.