Market Trends
Key Emerging Trends in the Pheochromocytoma Market
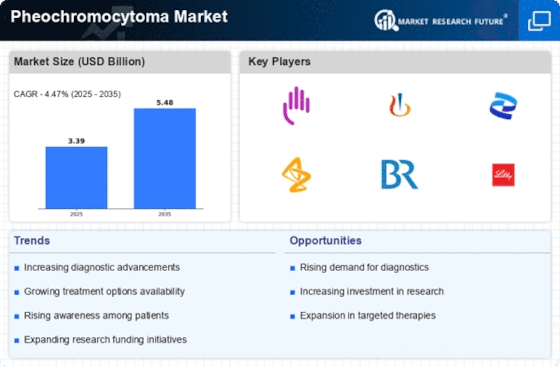
The market for pheochromocytoma, a rare neuroendocrine tumor affecting the adrenal glands, is witnessing notable trends in diagnostics, treatment, and awareness. As medical understanding of this condition advances, the market is adapting to address the unique challenges posed by pheochromocytoma and improve outcomes for affected individuals. One of the major innovative trend seen in the pheochromocytoma market is done by continued innovations in diagnostic imaging techniques. CT, MRI and PET are making early detection of pheochromocytoma more accurate with early intervention making the whole process prompt and strategic. Caused by a dominant autosomal inherited molecule pheochromocytoma has attracted an increased focus on genetic testing and screening, thus becoming crucial in the market. There are whole gene pools for which it is possible to preselect by identifying individuals with genetic predispositions such as families with syndromes like multiple endocrine neoplasia (MEN), or VHL syndrome; this helps to shape the landscape of preventive care. Globally, the market is in response to a very little rise in the prevalence of pheochromocytoma despite its incidence over the years being attributed to better diagnostic technology and increased understanding by medical practitioners. More awareness of such signs and symptoms ensuring proper and timely diagnosis improving illness outcome. There has been increasing reliance on use of alpha-blockers and beta-blockers as preoperative management to pheochromocytoma. These drugs provide the treatment management that effectively controls blood pressure and also prevents hypertensive crises during the surgical termination of tumor. The implementation of such pharmacological interventions is improving perioperative care tactics. The pheochromocytoma market is witnessing evolving postoperative monitoring strategies. Continuous blood pressure monitoring and cautious serological evaluations are the current tendency to exclude any residual tumor activity or recurrence, consolidating an all-inclusive strategy of treatment. The climate in pheochromocytoma medications is being defined by cooperative investigative endeavors and clinical preliminaries. The partnership of pharmaceutical firms, academic establishments, and healthcare organizations is promoting researches into discovery of new drugs to cure pheochromocytoma and future understanding of the molecular basis of the disease. In fact, the market is currently getting more attentions to the support groups for patients with pheochromocytoma. These groups help a lot in increasing awareness, availing resources and developing an environment that supports the patients and their families as a result, better overall patient well-being is achieved.
The rise of telemedicine is influencing the pheochromocytoma market by offering remote monitoring and virtual consultations. This trend enhances access to specialized care for patients in remote locations and facilitates ongoing communication between healthcare providers and individuals managing pheochromocytoma.



















Leave a Comment