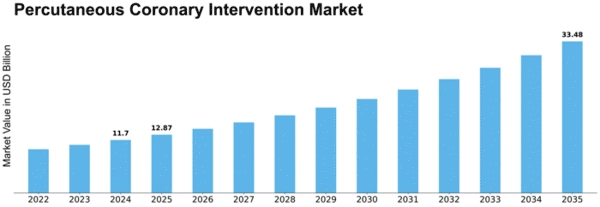
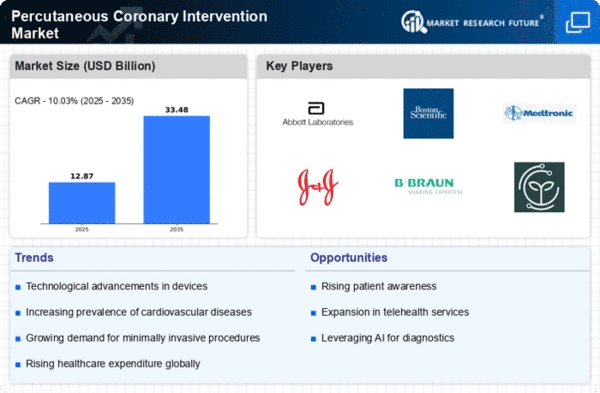
Percutaneous Coronary Intervention Size
Percutaneous Coronary Intervention Market Growth Projections and Opportunities
Percutaneous Coronary Intervention (PCI), a minimally invasive method for unblocking coronary arteries, is witnessing increased adoption driven by the growing incidence of heart diseases and a preference for less invasive procedures. Despite these positive trends, the market faces challenges due to infection risks. However, ongoing technological advancements present opportunities for global market leaders to further enhance PCI procedures, making them safer and more effective. The escalating prevalence of heart diseases worldwide has fueled the demand for PCI as an effective and less invasive treatment option. Patients and healthcare professionals alike are increasingly favoring minimally invasive procedures due to their associated benefits, including reduced recovery time and lower risk of complications.
While the market for PCI is on the rise, the potential threat of infections poses a challenge to its sustained growth. Infection risks associated with medical procedures are a concern that needs to be addressed to ensure the continued acceptance of PCI. Medical professionals and researchers are actively exploring ways to mitigate these risks and enhance the safety profile of PCI.
Despite the challenges, the PCI market holds promise, driven by continuous technological advancements. Innovations in medical technology are paving the way for enhanced PCI procedures, making them more precise, efficient, and safer for patients. The integration of cutting-edge technologies into PCI, such as improved imaging techniques and advanced catheter technologies, is expected to elevate the overall success and safety of these procedures.
Global market leaders in the medical field are well-positioned to capitalize on the opportunities presented by technological advancements in PCI. Collaborations, research initiatives, and investments in innovative solutions can contribute to the development of next-generation PCI procedures. This not only addresses existing challenges but also establishes a pathway for sustained market growth.
Collaborative efforts among healthcare professionals, researchers, and industry leaders play a crucial role in navigating the evolving landscape of PCI. By fostering partnerships and investing in research and development, the global medical community can address existing challenges, enhance patient safety, and drive continuous innovation in the field. This collaborative approach not only ensures the sustainability of the PCI market but also reinforces its position as a transformative solution in the realm of cardiac interventions.


















Leave a Comment