Market Trends
Key Emerging Trends in the Particle Therapy Market
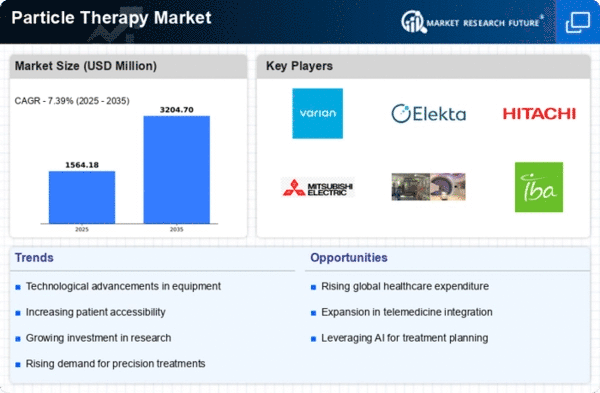
Recently, the particle therapy market has been experiencing significant movements. These trends are driven by technological advancement, rising cancer prevalence, and a growing awareness of the benefits offered by this novel treatment method. One key market trend is that the use of particle therapy as the preferred option for various cancer types is increasing at an alarming rate. In delivering high doses to tumors located near critical organs, traditional radiation therapy methods may be challenging. However, particle therapy addresses this challenge by allowing more focused targeting, hence reducing the risks of affecting healthy tissues around them. Thus, healthcare providers are currently incorporating particle therapy in their cancer treatment plans, thus contributing to the growth of the market. In addition, technology continues to shape particle therapy market dynamics considerably. Ongoing research and development efforts have resulted in advanced particle therapy systems with improved precision, efficiency, and patient outcomes, among other aspects. Some of these advances consist of real-time imaging during treatment, which enhances the accuracy of tumor targeting. Moreover, these market trends illustrate a rise in strategic partnerships and alliances between major companies related to the particle therapies industry that exist today. Collaboration between healthcare providers and research agencies with key players within the industry facilitates the sharing of expertise, resources, and technology. It also accelerates the development and implementation of new solutions for particle therapies, therefore making it more comprehensive and accessible in terms of cancer treatments. Furthermore, an increase in global cancer incidences significantly drives demand for particle therapy as a market player. The surge in cases has necessitated the need for sophisticated therapeutic interventions that can achieve effective results against cancers, which are becoming incisive now and then due to several factors, such as environmental pollution or lifestyle factors, among others. The targeted nature and precision involved when using particle therapies make it an attractive option for both patient and caregivers' consideration, particularly those who regard cost implications prior to diagnosis. Nonetheless, until recently, there were issues about cost-effectiveness, which hindered the spread of particle therapy. However, there seems to be a shift in these trends over the past few years. The increasing maturity and diffusion of technology have resulted in more emphasis on expenses and reimbursement models applied in the industry. Thus, streamlining workflow, improving treatment efficiency, and reducing equipment costs will make this technique more economically feasible, thereby increasing its acceptance by the market.

















Leave a Comment