Market Analysis
In-depth Analysis of Over-the-wire Micro-Guide Catheter Market Industry Landscape
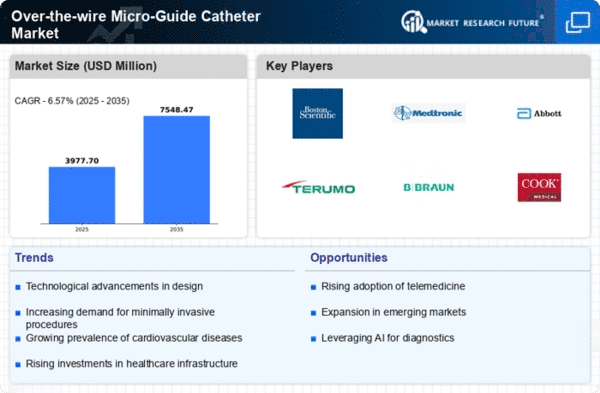
Growing Demand : The market for over-the-wire micro-guide catheters is witnessing a steady rise in demand due to the increasing prevalence of cardiovascular diseases and the rising adoption of minimally invasive procedures.
Technological Advancements : Continuous technological advancements in micro-guide catheters, such as improvements in material composition, design, and maneuverability, are driving market growth by enhancing procedural efficiency and patient outcomes.
Expanding Applications : The versatility of over-the-wire micro-guide catheters in various interventional procedures, including coronary angioplasty, neurovascular interventions, and peripheral vascular interventions, is expanding their market potential across different medical specialties.
Rising Healthcare Expenditure : The growing healthcare expenditure globally, coupled with increasing investments in healthcare infrastructure and facilities, is fueling the adoption of advanced medical devices like over-the-wire micro-guide catheters.
Market Competition : The market for over-the-wire micro-guide catheters is characterized by intense competition among key players, leading to constant innovations, strategic collaborations, and product launches aimed at gaining a competitive edge and expanding market share.
Regulatory Landscape : Stringent regulatory requirements and approval processes for medical devices pose challenges for market players, but adherence to quality standards is crucial for maintaining consumer trust and market credibility.
Market Consolidation : Mergers and acquisitions are prevalent in the over-the-wire micro-guide catheter market as companies seek to strengthen their product portfolios, expand their geographical presence, and leverage synergies to achieve sustainable growth.
Cost Containment Efforts : Healthcare cost containment efforts by governments and insurers may pose a challenge to market growth, as pricing pressures and reimbursement policies influence purchasing decisions and market dynamics.
Emerging Markets : The market for over-the-wire micro-guide catheters is witnessing significant growth opportunities in emerging markets due to rising healthcare infrastructure development, increasing disposable income levels, and a growing patient pool with unmet medical needs.
Patient Safety and Comfort : Focus on enhancing patient safety and comfort during interventional procedures is driving innovations in over-the-wire micro-guide catheter design, with features aimed at reducing procedural risks and improving patient experience.
Educational Initiatives : Educational initiatives and training programs aimed at healthcare professionals are essential for promoting awareness about the benefits and proper usage of over-the-wire micro-guide catheters, thus driving market growth through increased adoption and procedural expertise.


















Leave a Comment