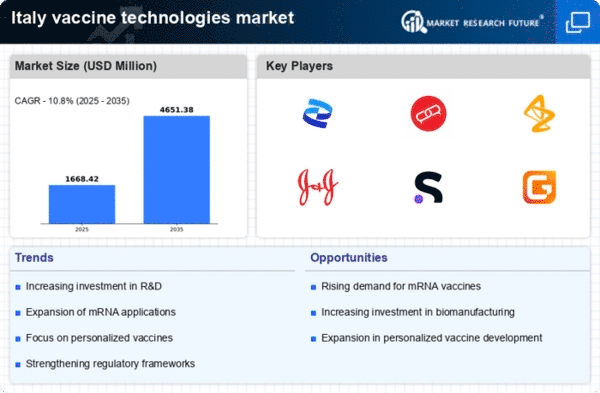
Italy Security, Access Control and Robotics Market Segmentation
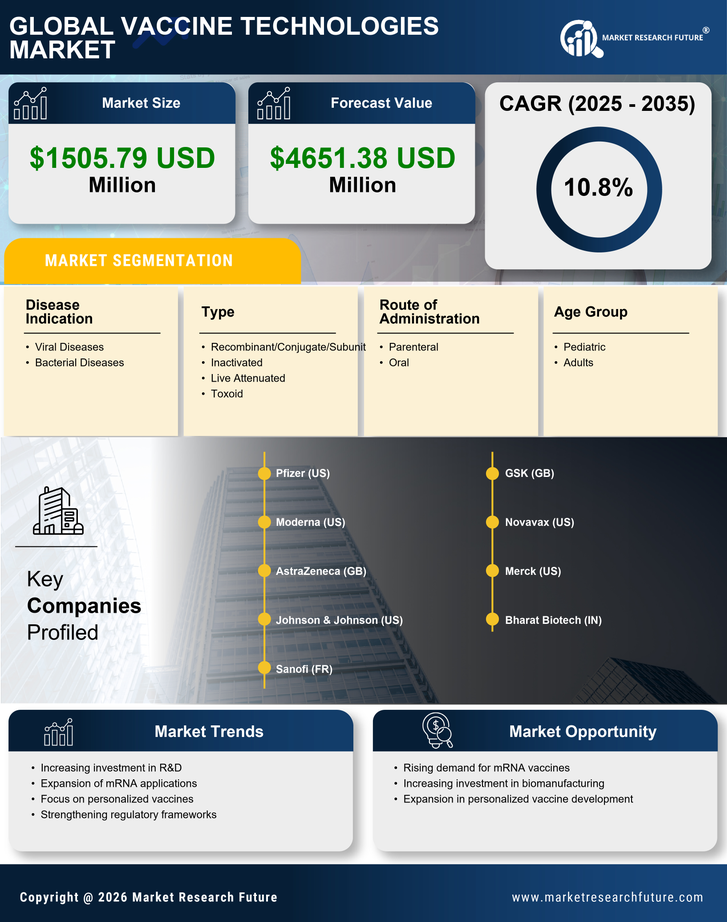
Security, Access Control and Robotics By Type (USD Million, 2025-2035)
- Recombinant/Conjugate/Subunit
- Inactivated
- Live Attenuated
- Toxoid
Security, Access Control and Robotics By Route of Administration (USD Million, 2025-2035)
- Parenteral
- Oral
Security, Access Control and Robotics By Disease Indication (USD Million, 2025-2035)
- Viral Diseases
- Bacterial Diseases
Security, Access Control and Robotics By Age Group (USD Million, 2025-2035)
- Pediatric
- Adults
Security, Access Control and Robotics By Distribution Channel (USD Million, 2025-2035)
- Hospital & Retail Pharmacies
- Government Suppliers
- Others