Inhaled Nitric Oxide No Delivery System Market Summary
As per Market Research Future analysis, the Inhaled Nitric Oxide No Delivery System Market. Size was estimated at 1.268 USD Billion in 2024. The Inhaled Nitric Oxide No Delivery System industry is projected to grow from 1.354 USD Billion in 2025 to 2.619 USD Billion by 2035, exhibiting a compound annual growth rate (CAGR) of 6.82% during the forecast period 2025 - 2035
Key Market Facts 2025-2035
| Metric | 2025 Value | 2035 Projection | CAGR |
|---|---|---|---|
| Global Market Size | USD 1.354B | USD 2.619B | 6.82% |
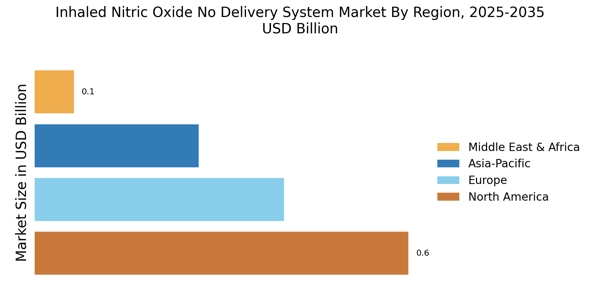
| North America | USD 542M (40%) | USD 1.048B | 6.8% ← DOMINANT |
| Asia Pacific | USD 406M (30%) | USD 786M | 6.9% ← FASTEST |
| Europe | USD 271M (20%) | USD 524M | 6.7% |
| Concentration | 2025 Share | CAGR | Key Application |
|---|---|---|---|
| 300 ppm | 42.1% | 7.4% | DOMINANT - Adult PH/ARDS rescue |
| 200 ppm | 28.6% | 7.8% | FASTEST - Neonatal transition |
| 100 ppm | 18.4% | 6.5% | PPHN standard |
| Delivery System | 2025 Share | FiO₂ Range | Patient Group |
|---|---|---|---|
| Ventilator | 68.4% | 21-100% | NICU/ICU (±0.1ppm precision) |
| Nasal Cannula | 19.2% | 24-44% | Non-invasive neonates |
| Face Mask | 7.5% | 35-60% | Pediatrics |
| Tracheal Tube | 4.9% | 100% | Surgical |
| Population | 2025 Share | Incidence |
|---|---|---|
| Newborns | 62.3% | 1:1,000 births (PPHN) |
| Adults | 22.1% | PH/ARDS |
| Children | 10.6% | Ped ARDS |
| Older Adults | 5.0% | COPD/PH |
| Indication | 2025 Share | Evidence Level |
|---|---|---|
| Pulmonary Hypertension | 48.7% | FDA/EMA approved |
| Respiratory Distress Syndrome | 28.4% | Neonatal standard |
| COPD/Asthma | 22.9% | Emerging |
| End User | 2025 Share | Capacity |
|---|---|---|
| Hospitals | 82.6% | NICU/ICU dominance |
| Clinics/ASC | 17.4% | Outpatient PH |
North America (40%): Largest market, advanced NICUs, INOmax® leadership
Asia Pacific (30%): Fastest growing, preterm birth surge (15M/year), urbanization
Europe (20%): EMA guidelines, national health systems
Primary Growth Drivers
Technological advancements (+3.2% CAGR): Portable/tankless systems
Non-invasive treatments (+2.1% CAGR): Nasal cannula adoption
Respiratory disorder prevalence (+1.5% CAGR): PPHN, ARDS, COPD
Pulmonary care advances (+0.9% CAGR): 300ppm protocols
Key Market Trends & Highlights
The Inhaled Nitric Oxide No Delivery System Market is poised for growth driven by technological advancements and increasing demand for non-invasive treatments.
- Technological advancements are enhancing the efficacy and safety of inhaled nitric oxide delivery systems.
- North America remains the largest market, while Asia-Pacific is emerging as the fastest-growing region in this sector.
- The 300 ppm segment dominates the market, whereas the 200 ppm segment is witnessing rapid growth due to its innovative applications.
- Rising prevalence of respiratory disorders and growing awareness of non-invasive treatment options are key drivers propelling market expansion.
Market Size & Forecast
| 2024 Market Size | 1.268 (USD Billion) |
| 2035 Market Size | 2.619 (USD Billion) |
| CAGR (2025 - 2035) | 6.82% |
Major Players
Mallinckrodt Pharmaceuticals (IE), Bellerophon Therapeutics (US), United Therapeutics Corporation (US), OxySure Therapeutics (US), AstraZeneca (GB), Pfizer Inc. (US), Novartis AG (CH), Eli Lilly and Company (US)
.png?v=1776933543)