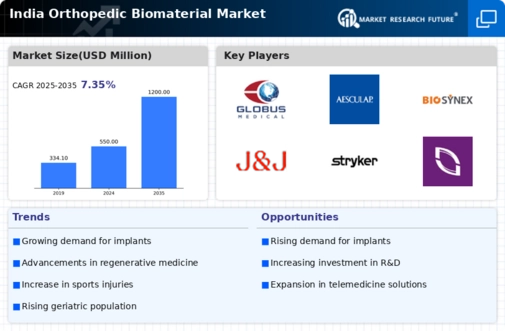
India Industrial Automation & Equipment Market Segmentation
Industrial Automation & Equipment By Type (USD Million, 2025-2035)
- Ceramics
- Polymers
- Composites
- Metals
Industrial Automation & Equipment By Application (USD Million, 2025-2035)
- Bone Grafting
- Joint Reconstruction
- Spinal Fusion
- Fracture Repair
Industrial Automation & Equipment By End User (USD Million, 2025-2035)
- Hospitals
- Orthopedic Clinics
- Ambulatory Surgical Centers
Industrial Automation & Equipment By Form (USD Million, 2025-2035)
- Granules
- Putty
- Sheets
- Blocks