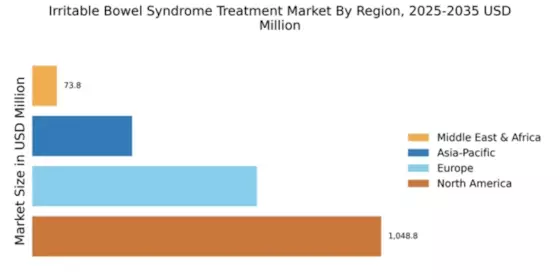
North America : Market Leader in IBS Treatment
North America is poised to maintain its leadership in the Irritable Bowel Syndrome (IBS) treatment market, holding a significant share of approximately 1048.77 million in 2024. The region's growth is driven by increasing awareness of IBS, advancements in treatment options, and supportive regulatory frameworks. The demand for innovative therapies is further fueled by a rising prevalence of IBS, with healthcare policies promoting better access to treatment. The competitive landscape in North America is robust, featuring key players such as AbbVie, Pfizer, and Bristol-Myers Squibb. These companies are actively engaged in research and development to introduce novel therapies. The U.S. remains the largest market, supported by a well-established healthcare infrastructure and a high level of investment in pharmaceutical innovations. This dynamic environment positions North America as a critical hub for IBS treatment advancements.
Europe : Emerging Market for IBS Solutions
Europe is witnessing a growing demand for Irritable Bowel Syndrome (IBS) treatments, with a market size of approximately 675.0 million in 2024. Factors such as increasing healthcare expenditure, a focus on patient-centered care, and the rising incidence of IBS are driving this growth. Regulatory bodies are also playing a crucial role in facilitating the approval of new therapies, enhancing patient access to effective treatments. Leading countries in this region include Germany, France, and the UK, where major pharmaceutical companies like Boehringer Ingelheim and GlaxoSmithKline are actively involved. The competitive landscape is characterized by a mix of established players and emerging biotech firms, fostering innovation in treatment options. The European market is expected to expand further as healthcare policies increasingly prioritize digestive health.
Asia-Pacific : Rapidly Growing IBS Market
The Asia-Pacific region is emerging as a significant player in the Irritable Bowel Syndrome (IBS) treatment market, with a market size of around 300.0 million in 2024. The growth is driven by increasing awareness of IBS, rising disposable incomes, and a growing focus on healthcare infrastructure. Additionally, the region's diverse population presents unique challenges and opportunities for tailored treatment solutions, further propelling market expansion. Countries like Japan, China, and India are at the forefront of this growth, with key players such as Takeda Pharmaceutical Company leading the charge. The competitive landscape is evolving, with both multinational corporations and local firms striving to capture market share. As the region continues to invest in healthcare advancements, the IBS treatment market is expected to flourish, addressing the needs of a growing patient population.
Middle East and Africa : Developing Market with Challenges
The Middle East and Africa (MEA) region is gradually emerging in the IBS treatment market, currently valued at $73.76M in 2024. The Middle East and Africa region is gradually emerging in the irritable bowel syndrome treatment market. Growth opportunities are increasingly visible in emerging healthcare ecosystems including the gcc irritable bowel syndrome treatment market. The growth is driven by increasing healthcare investments, a rising prevalence of IBS, and a growing awareness of digestive health. However, challenges such as limited access to healthcare facilities and varying regulatory environments can hinder market growth. Governments are beginning to recognize the importance of gastrointestinal health, which may lead to improved healthcare policies in the future. Countries like South Africa and the UAE are at the forefront of this market, with increasing investments in healthcare infrastructure. The presence of key players is limited, but companies are beginning to explore opportunities in this region. As awareness of IBS grows, the demand for effective treatments is expected to rise, presenting significant growth potential for the market.