Halal Pharmaceuticals Market
ID: MRFR/Pharma/38947-HCR
100 Pages
Rahul Gotadki
Last Updated: April 06, 2026
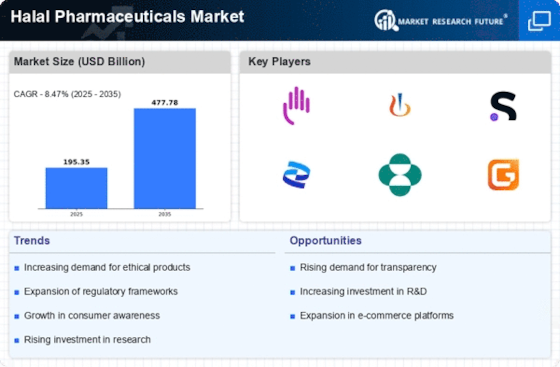
Halal Pharmaceuticals Market Research Report By Drug Type (Prescription Drugs, Over-the-Counter (OTC) Drugs, Nutraceuticals, Cosmetics, Medical Devices), By Therapeutic Area (Cardiovascular Diseases, Diabetes, Anti-Infectives, Gastrointestinal Disorders, Respiratory Diseases, Oncology, Neurological Disorders, Others), By Dosage Form (Tablets and Capsules, Injectables, Topicals, Orals, Others), By End-User (Hospitals and Clinics, Pharmacies, Retailers, Online Pharmacies, Others), By Certification (Halal International Standard, Malaysia Standard, Gulf Standard, Others) and By Regional - Growth & Industry Forecast 2025 To 2035