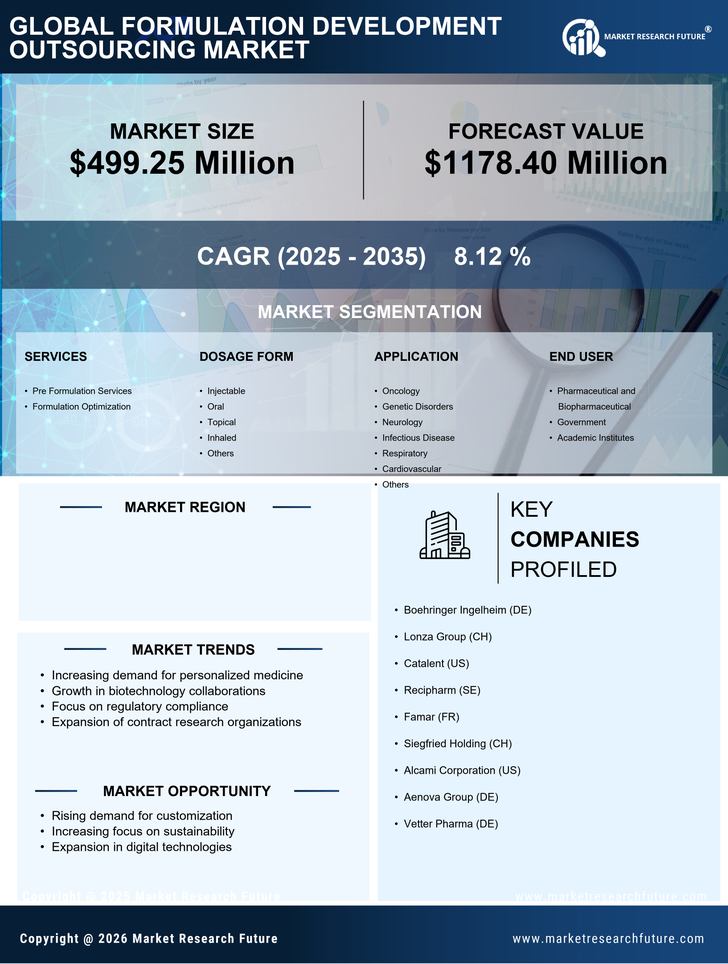
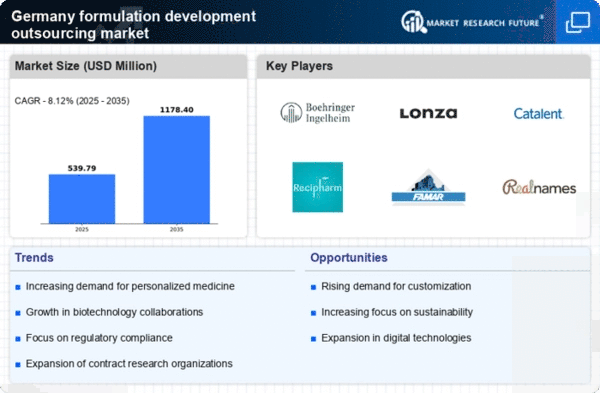
Germany Pharmaceutical Market Segmentation
Pharmaceutical By Services (USD Million, 2025-2035)
- Pre Formulation Services
- Formulation Optimization
Pharmaceutical By Dosage Form (USD Million, 2025-2035)
- Injectable
- Oral
- Topical
- Inhaled
- Others
Pharmaceutical By Application (USD Million, 2025-2035)
- Oncology
- Genetic Disorders
- Neurology
- Infectious Disease
- Respiratory
- Cardiovascular
- Others
Pharmaceutical By End User (USD Million, 2025-2035)
- Pharmaceutical and Biopharmaceutical
- Government
- Academic Institutes