North America : Leading Market for Innovation
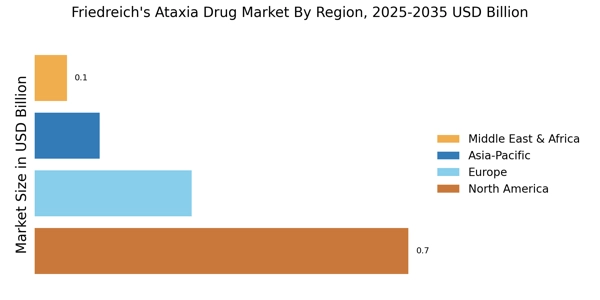
North America leads in the Friedreich's Ataxia Drug Market, accounting for over 60% of the global revenue in 2024. The region benefits from advanced healthcare infrastructure, significant R&D investments, and a growing patient population. Regulatory support from agencies like the FDA accelerates drug approvals, fostering innovation and enhancing treatment accessibility. The increasing prevalence of Friedreich's Ataxia is driving demand for effective therapies, further propelling market growth.
The United States holds approximately 48% share, valued at USD 0.55 Billion, driven by strong orphan drug development and advanced clinical research, while Canada accounts for 12% share, valued at USD 0.14 Billion, supported by growing rare disease funding.
The United States is the primary contributor to this market, with key players such as Reata Pharmaceuticals, Pfizer, and Bristol-Myers Squibb leading the charge. The competitive landscape is characterized by ongoing clinical trials and collaborations aimed at developing novel therapies. The presence of established pharmaceutical companies ensures a robust pipeline of potential treatments, enhancing the region's position as a hub for Friedreich's Ataxia drug development.
Europe : Emerging Market with Potential
Europe Friedreich's Ataxia Drug Market size was valued at USD 0.29 billion in 2024, making it the second-largest regional market with a 25% share. The region is witnessing a surge in demand driven by increasing awareness of the disease and advancements in treatment options. Regulatory bodies like the European Medicines Agency (EMA) are actively facilitating the approval of new therapies, which is crucial for market expansion. The growing collaboration between public and private sectors is also a significant catalyst for growth in this region.
Germany holds 8% share valued at USD 0.09 Billion, driven by strong pharmaceutical innovation, while the United Kingdom accounts for 7% share valued at USD 0.08 Billion, supported by increasing genetic research initiatives.
Leading countries in Europe include Germany, France, and the United Kingdom, where the presence of major pharmaceutical companies like Sanofi and Bayer enhances the competitive landscape. The market is characterized by a mix of established players and emerging biotech firms, all striving to innovate and provide effective treatments. The focus on patient-centric approaches and personalized medicine is shaping the future of Friedreich's Ataxia therapies in Europe.
Asia-Pacific : Emerging Powerhouse for Treatments
Asia-Pacific is rapidly emerging as a significant market for Friedreich's Ataxia drugs, currently holding about 10% of the global market share. The region's growth is driven by increasing healthcare investments, rising awareness of rare diseases, and a growing patient population. Countries like Japan and Australia are leading the charge, supported by favorable regulatory environments that encourage drug development and approval. The increasing prevalence of Friedreich's Ataxia is also contributing to the demand for effective therapies.
China holds 4% share valued at USD 0.05 Billion, driven by expanding biotech capabilities, while Japan accounts for 3% share valued at USD 0.03 Billion, supported by advanced healthcare infrastructure.
Japan stands out as a key player in this market, with companies like Astellas Pharma actively involved in research and development. The competitive landscape is evolving, with both local and international firms vying for market share. Collaborations between biotech firms and research institutions are fostering innovation, while government initiatives aimed at improving healthcare access are further enhancing the market's potential in the Asia-Pacific region.
Middle East and Africa : Untapped Market Opportunities
The Middle East and Africa region represents an emerging market for Friedreich's Ataxia drugs, currently accounting for about 5% of the global market share. The growth in this region is primarily driven by increasing healthcare investments and a rising awareness of rare diseases. However, challenges such as limited access to healthcare and regulatory hurdles remain. Governments are beginning to recognize the importance of addressing rare diseases, which is expected to catalyze market growth in the coming years.
The UAE holds 2% share valued at USD 0.02 Billion, driven by healthcare investments, while South Africa accounts for 1.5% share valued at USD 0.017 Billion, supported by improving rare disease awareness.
Countries like South Africa and the United Arab Emirates are at the forefront of this market, with efforts to improve healthcare infrastructure and access to treatments. The competitive landscape is still developing, with a mix of local and international players entering the market. As awareness grows and regulatory frameworks improve, the potential for Friedreich's Ataxia drug development in the Middle East and Africa is significant, paving the way for future opportunities.