Embolic Protection Devices Market Summary
As per MRFR analysis, the Embolic Protection Devices Market Size was estimated at 357.73 USD Million in 2024. The Embolic Protection Devices industry is projected to grow from 387.78 USD Million in 2025 to 868.89 USD Million by 2035, exhibiting a compound annual growth rate (CAGR) of 8.4% during the forecast period 2025 - 2035.
Key Market Trends & Highlights
The Embolic Protection Devices Market is poised for substantial growth driven by technological advancements and increasing cardiovascular disease prevalence.
- Technological advancements are enhancing the efficacy and safety of embolic protection devices, thereby attracting greater market interest.
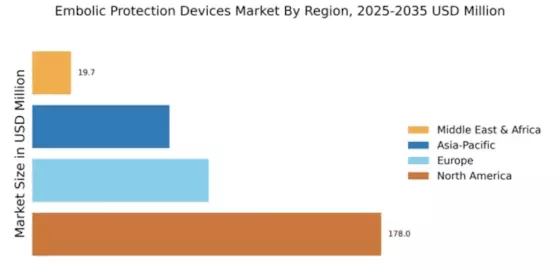
- North America remains the largest market for embolic protection devices, while the Asia-Pacific region is recognized as the fastest-growing market.
- In the cardiovascular surgery segment, embolic protection devices are widely utilized, whereas the transcatheter aortic valve replacement segment is experiencing rapid growth.
- The increasing prevalence of cardiovascular diseases and regulatory support for innovative devices are key drivers propelling market expansion.
Market Size & Forecast
| 2024 Market Size | 357.73 (USD Million) |
| 2035 Market Size | 868.89 (USD Million) |
| CAGR (2025 - 2035) | 8.4% |
Major Players
Medtronic(US), Boston Scientific (US), Abbott (US), Terumo (JP), Cardinal Health (US), B. Braun (DE), Johnson & Johnson (US), Stryker (US), Cook Medical (US)