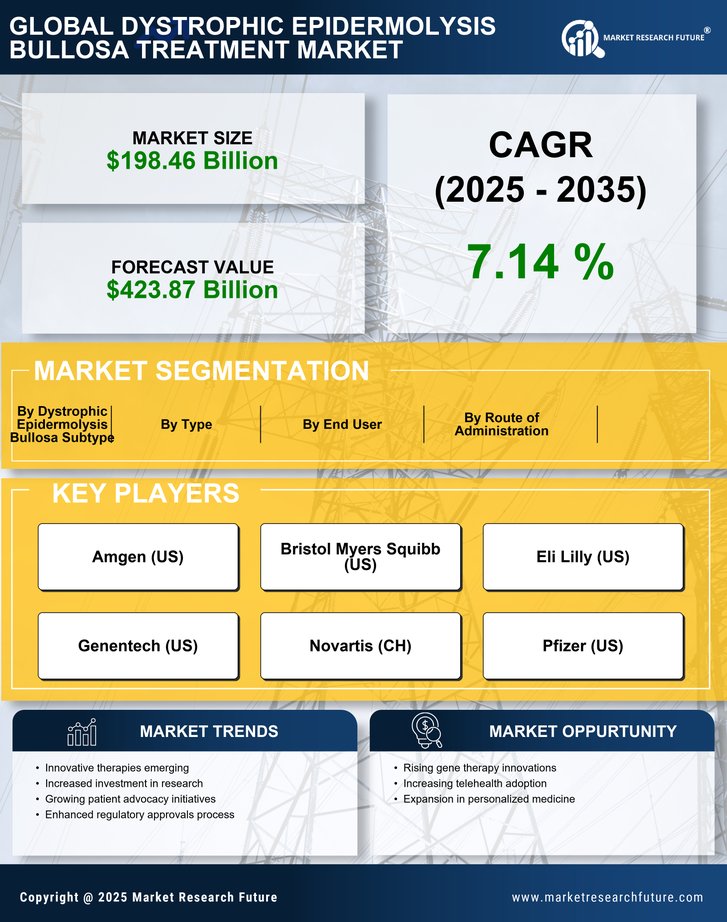
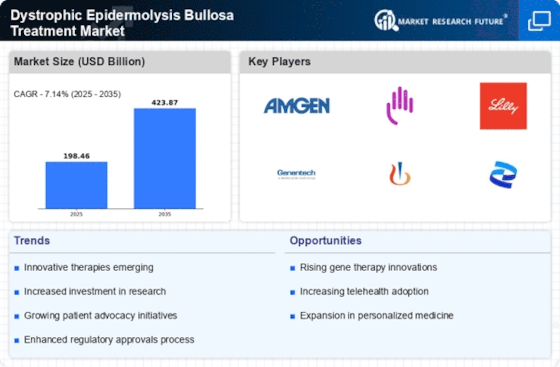
Healthcare Market Segmentation
Healthcare By Type (USD Billion, 2025-2035)
- Initial Treatment
- Palliative Treatment
- Surgical Treatment
Healthcare By Route of Administration (USD Billion, 2025-2035)
- Oral
- Topical
- Injectables
Healthcare By End User (USD Billion, 2025-2035)
- Hospitals
- Clinics
- Homecare Settings
Healthcare By Dystrophic Epidermolysis Bullosa Subtype (USD Billion, 2025-2035)
- EBS Simplex
- EBS Dystrophica
- EBS Junctional