Companion Diagnostic Market Summary
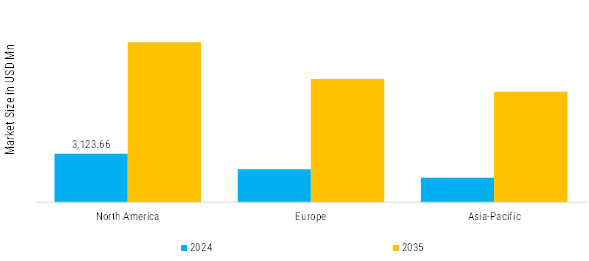
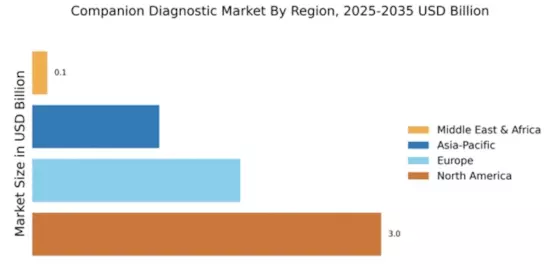
According to Market Research Future analysis, the Companion Diagnostics Market size was valued at USD 6,828.25 million in 2024. The market is projected to grow from USD 7,694.07 million in 2025 to USD 25,300.58 million by 2035, exhibiting a CAGR of 12.68% during the forecast period 2025–2035. North America led the market with over 0.04% share, generating around USD 3 million in revenue.
Rising global cancer burden and expanding precision medicine adoption are driving companion diagnostics demand. Increasing biomarker-based treatment decisions, targeted therapy approvals, and personalized oncology approaches are accelerating adoption of companion diagnostics across clinical workflows and drug development pipelines worldwide.
- According to the World Health Organization (WHO), nearly 20 million new cancer cases and 9.7 million deaths occurred globally in 2022, while 1 in 5 individuals develop cancer during their lifetime, significantly increasing demand for biomarker-based testing and companion diagnostics supporting personalized treatment decisions.
Key Market Trends & Highlights
The Companion Diagnostics Market is currently valued in the high single to low double digit billions of dollars and is expanding rapidly, underpinned by the rise of precision medicine, oncology targeted therapies, and biomarker driven drug development.
- North America leads Companion Diagnostics Market, accounting for over 0.04% global revenue in 2024.
- PCR technology captured 36% market share, driven by high accuracy and cost-effective testing adoption.
- Assays, reagents & kits segment held 44% share, supported by growing biomarker testing demand.
- Cancer indication dominated market with 63% share in 2024, driven by increasing global cancer burden.
Market Size & Forecast
| 2024 Market Size | 6,828.25 (USD Million) |
| 2035 Market Size | 25,300.58 (USD Million) |
| CAGR (2025 - 2035) | 12.68% |
Major Players
Companies such as Qiagen, Bio-Techne, BioMérieux, Thermo Fisher Scientific Inc., Illumina, Inc., Agilent Technologies, Inc., F. Hoffmann-la Roche Ltd. are some of the major participants in the global market.