Clinical Research and Trial Consulting Services Market
ID: MRFR/PS/64414-HCR
200 Pages
Rahul Gotadki
Last Updated: March 28, 2026
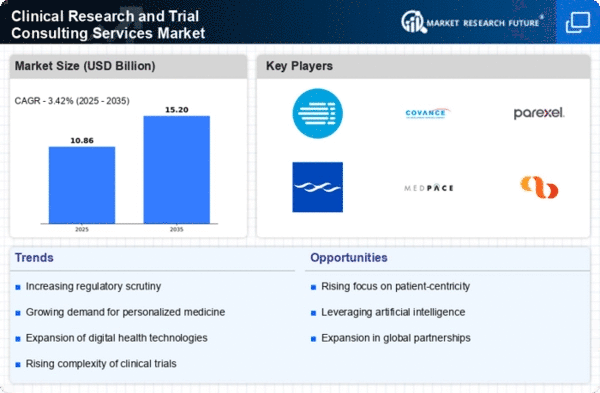
Clinical Research and Trial Consulting Services Market Research Report Information By End Use (Pharmaceutical Companies, Biotechnology Firms, Medical Device Manufacturers, Academic Institutions), By Application (Clinical Trial Management, Regulatory Affairs Consulting, Data Management Services, Biostatistics Services), By Study Phase (Preclinical Phase, Phase I, Phase Ii, Phase Iii), By Service Type (Consulting Services, Project Management Services, Monitoring Services, Training Services), By Therapeutic Area (Oncology, Cardiology, Neurology, Infectious Diseases) And By Region (North America, Europe, Asia-Pacific, And Rest Of The World) – Market Forecast Till 2035.