Carcinoembryonic Antigen Market Summary
As per MRFR analysis, the Carcinoembryonic Antigen Market Size was estimated at 1451.77 USD Million in 2024. The Carcinoembryonic Antigen industry is projected to grow from 1672.74 USD Million in 2025 to 6897.88 USD Million by 2035, exhibiting a compound annual growth rate (CAGR) of 15.22% during the forecast period 2025 - 2035.
Key Market Trends & Highlights
The Carcinoembryonic Antigen Market is poised for substantial growth driven by technological advancements and increasing demand for personalized medicine.
- Technological advancements in diagnostics are enhancing the accuracy and efficiency of Carcinoembryonic Antigen testing.
- The shift towards personalized medicine is fostering tailored treatment approaches, thereby increasing the relevance of biomarkers like Carcinoembryonic Antigen.
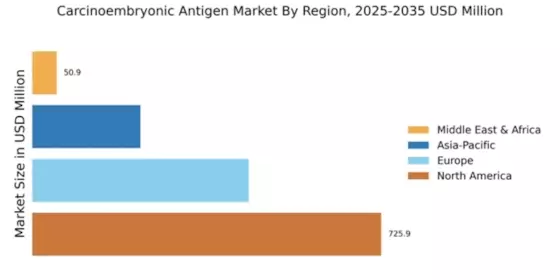
- North America remains the largest market, while the Asia-Pacific region is emerging as the fastest-growing area for Carcinoembryonic Antigen applications.
- Rising incidence of cancer and advancements in diagnostic technologies are key drivers propelling market growth.
Market Size & Forecast
| 2024 Market Size | 1451.77 (USD Million) |
| 2035 Market Size | 6897.88 (USD Million) |
| CAGR (2025 - 2035) | 15.22% |
Major Players
Roche (CH), Abbott Laboratories (US), Thermo Fisher Scientific (US), Siemens Healthineers (DE), Bristol-Myers Squibb (US), Merck KGaA (DE), Johnson & Johnson (US), F. Hoffmann-La Roche AG (CH)