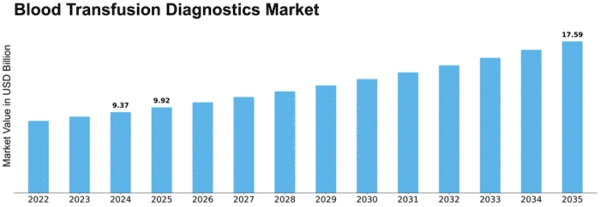
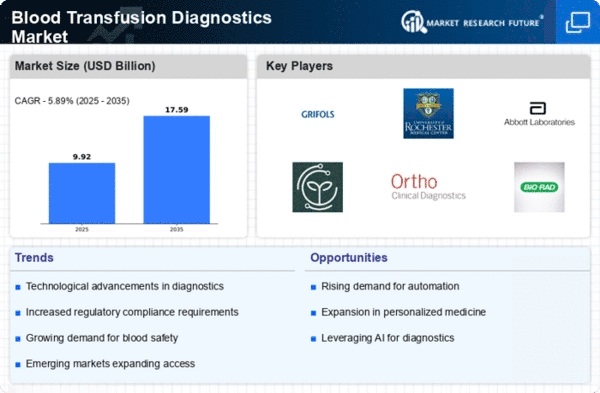
Blood Transfusion Diagnostics Size
Blood Transfusion Diagnostics Market Growth Projections and Opportunities
Since blood transfusions are needed worldwide, the blood transfusion diagnostics industry is growing. Many factors contribute to the growth in blood transfusions. Surgical operations, trauma, and anemia are examples. This issue has driven demand for diagnostic equipment and testing. The blood screening sector has been greatly impacted by technological advances. Blood transfusion diagnoses are improved by sophisticated screening technologies including nucleic acid testing (NAT) and enhanced immunoassays. This is done to fulfill the growing need for safe, trustworthy blood products. The market is affected by strict blood safety and transfusion diagnostics regulations. This is the result of constraints. Since regulatory organizations worldwide have set strict standards for screening given blood for infectious illnesses, the usage of modern diagnostic methods that meet these requirements has increased. Blood-donating organizations must undergo extensive screening due to the presence of blood-borne infections including HIV, hepatitis, and others. Diagnostics for blood transfusions are crucial because they identify and eliminate tainted blood units. This shields blood donors and receivers from health dangers. The market is growing due to increased awareness of safe blood transfusions and diagnostics' role in blood safety. This understanding helps keep blood safe. Healthcare institutions and government organizations are promoting transfusion diagnostic technology via education. Healthcare facilities run these programs. Blood supply problems, exacerbated by natural catastrophes and public health issues, highlight the need for effective and efficient blood transfusion diagnostics. Diagnostic tools that assure safe blood product availability amid shortages are growing in popularity. These technologies accelerate screening. Diagnostic technologies in healthcare systems are a major market driver. Automated and integrated transfusion diagnostics systems help hospitals and blood banks streamline blood transfusion services and testing. These systems improve testing efficiency. The amount of blood given is rising worldwide due to voluntary blood donation campaigns. Effective, high-throughput blood transfusion diagnostics will be needed as donor numbers grow. This will boost market growth. International blood transfusion disease control methods are changing the business world. WHO is leading blood safety programs. Healthcare institutions must invest in advanced transfusion diagnostics to screen effectively, according to the WHO. WHO leads these activities. The COVID-19 pandemic has affected the Blood Transfusion Diagnostics Market by highlighting the necessity for a robust and secure blood supply chain. Modern diagnoses have been hastened due to pandemic measures and screening. These procedures protect blood donors and recipients.



















Leave a Comment