Market Analysis
In-depth Analysis of Blood Transfusion Diagnostics Market Industry Landscape
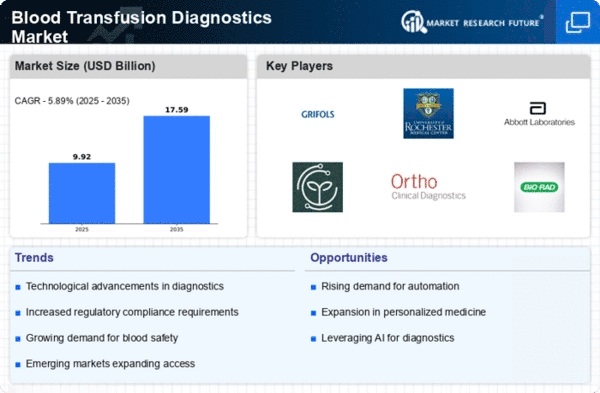
Increasing surgical procedures, trauma, and chronic illnesses are driving blood transfusion demand. This is really crucial. The increased demand for blood components requires effective diagnostic tools to assure safe and compatible blood transfusions. Technology innovations shape market dynamics. Diagnostic accuracy and efficiency have improved due to blood type, cross-matching, and nucleic acid testing advances. Continuous improvements enabled these advances. Because of this, blood transfusions are safer, which has driven industry expansion. The industry is affected by strict blood safety and transfusion regulations. Thus, the industry is heavily influenced. As a need, blood transfusion diagnostics companies must follow rules. This is because regulatory limits affect product development, testing, and marketing plans for new markets. A regular supply of donors is needed to ensure safe blood availability. An increased and diversified blood supply due to blood donation and awareness initiatives has increased the need for sophisticated diagnostics to match and screen blood. The more diversified blood supply has caused this. Standardized worldwide blood transfusion diagnoses are needed as healthcare systems become more interconnected. Because of this predisposition, uniform testing methodologies have been created, allowing market participants to create widely accepted solutions. Globally, hemophilia and anemia are on the increase. This has increased the need for reliable blood transfusion testing to assure compatibility and minimize adverse reactions. Thus, market dynamics have been affected. Molecular diagnostics for blood transfusion recipient compatibility is becoming industry-wide. Due to its high sensitivity in identifying viral infections, nucleic acid testing (NAT) has become widespread. This has improved blood safety and modified customer desires. It is crucial to have high-quality, reliable diagnostics for blood transfusions. Quality assurance and standardization initiatives in the industry shape the competitive climate and testing process acceptability. These activities set the competitive scene. Diagnostic firms and healthcare institutions or blood banks are forming more relationships, which is projected to continue. These agreements simplify diagnosis, improve blood transfusion services, and foster market innovation. Affordability is a major issue in healthcare systems with budgetary constraints. To meet the unique economic landscapes in different regions, market participants are developing precise and competitive diagnostic solutions. This is done to fulfill economic requirements.



















Leave a Comment