Biological Safety Testing Products and Services Market Summary
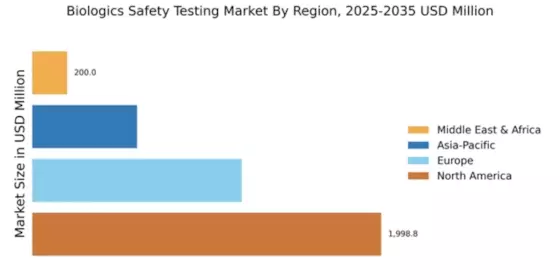
April 01, 2026- According to Market Research Future analysis, the Biologics Safety Testing Market Size was estimated at 3998.79 USD Million in 2024. The Biologics Safety Testing industry is projected to grow from 4542.71 USD Million in 2025 to 16262.06 USD Million by 2035, exhibiting a compound annual growth rate (CAGR) of 13.6% during the forecast period 2025 - 2035. North America led the market with over 49.97% share, generating around USD 1998.8 billion in revenue.
Rapid expansion of biologics, vaccines, and advanced therapies is driving demand for stringent safety testing. Increasing global immunization programs, complex biologic pipelines, and regulatory focus on contamination control significantly boost market growth across pharmaceutical and biotechnology sectors worldwide.
WHO reports immunization prevents 3.5–5 million deaths annually. Gavi has helped vaccinate over 1 billion children since 2000. UNICEF states global DTP3 coverage reached 84% (2023), increasing demand for biologics safety testing to ensure product quality and biosafety compliance.
Key Market Trends & Highlights
The Biologics Safety Testing Market is poised for substantial growth driven by technological advancements and regulatory compliance.
- North America dominates with 49.97% share in 2024, driven by advanced pharmaceutical infrastructure and strong regulatory compliance systems.
- Europe market valued at USD 1,199.65 million in 2024, accounting for 30% global share with strong EMA regulations.
- Sterility Testing holds largest application share at 32% in 2024, driven by strict contamination control in biologics production.
- Molecular assays grow fastest, expanding at 30% share due to advanced genomic technologies enabling rapid biologics safety evaluation.
Market Size & Forecast
| 2024 Market Size | 3998.79 (USD Million) |
| 2035 Market Size | 16262.06 (USD Million) |
| CAGR (2025 - 2035) | 13.6% |
Major Players
Companies such as Charles River Laboratories (US), Eurofins Scientific (FR), Sartorius AG (DE), Merck KGaA (DE), Thermo Fisher Scientific (US), Lonza Group (CH), WuXi AppTec (CN), SGS SA (CH), Boehringer Ingelheim (DE) are some of the major participants in the global market.