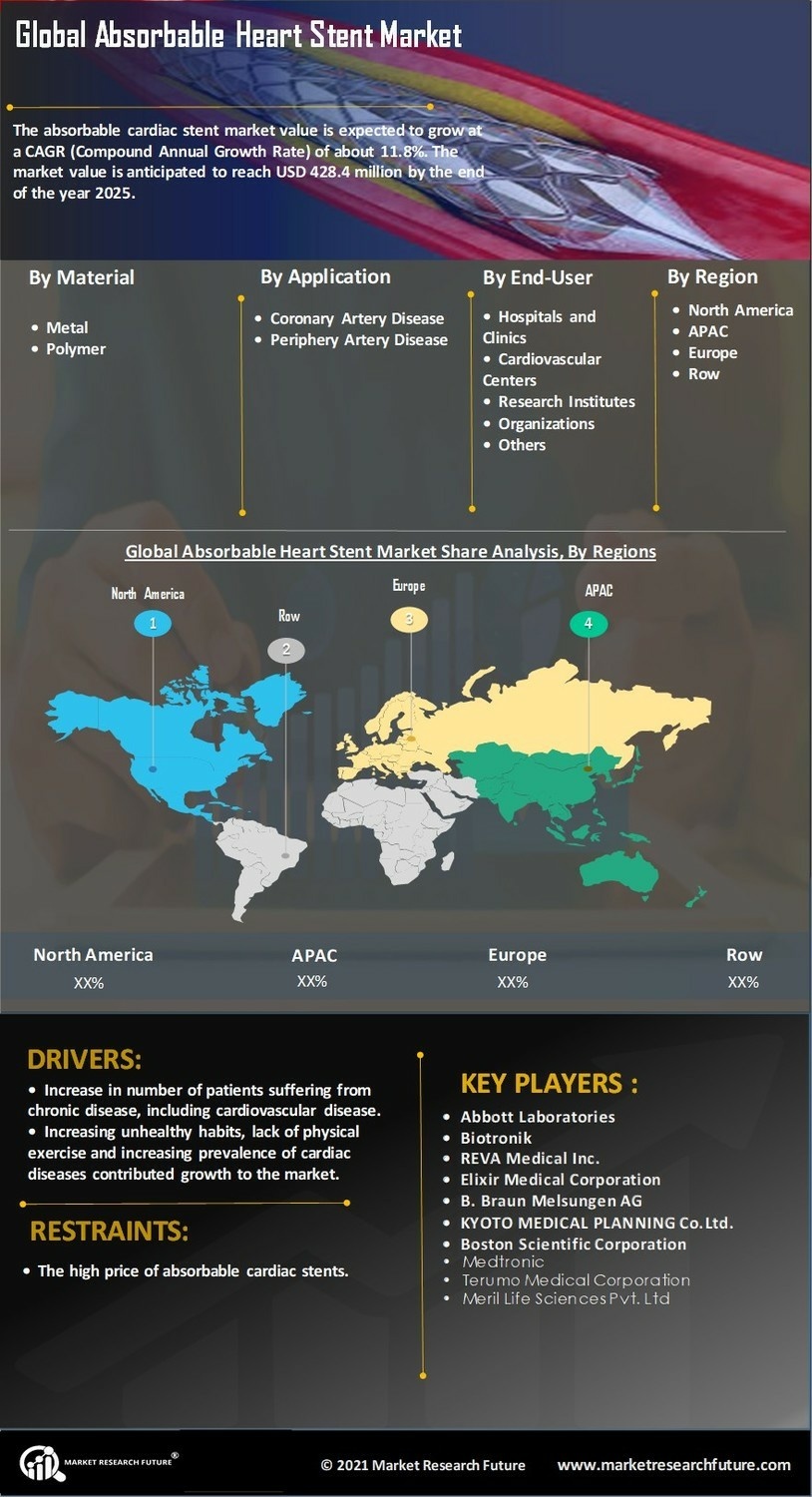
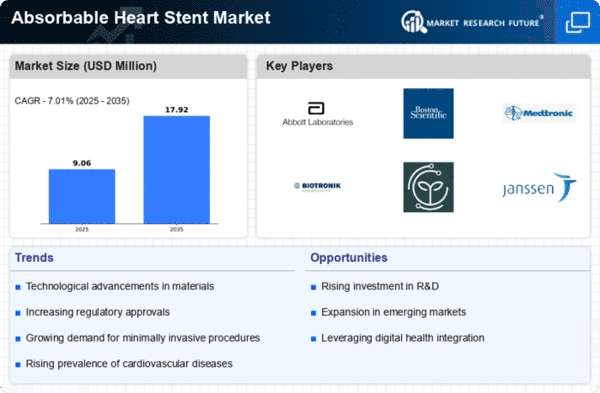
Healthcare Market Segmentation
Healthcare By Application (USD Million, 2025-2035)
- Coronary Artery Disease
- Peripheral Artery Disease
- Structural Heart Disease
- Congenital Heart Defects
Healthcare By Material Type (USD Million, 2025-2035)
- Polylactic Acid
- Polyglycolic Acid
- Polycaprolactone
- Composite Materials
Healthcare By End User (USD Million, 2025-2035)
- Hospitals
- Ambulatory Surgical Centers
- Cardiology Clinics
Healthcare By Delivery Method (USD Million, 2025-2035)
- Percutaneous Coronary Intervention
- Surgical Implantation
- Transcatheter Aortic Valve Replacement
Healthcare By Regulatory Approval Status (USD Million, 2025-2035)
- Approved
- Under Review
- Research Phase