Global market valuation was derived through a hybrid revenue-mapping and patient-volume estimation methodology. The analytical framework included:
Manufacturer Mapping: Identification of 35+ key biosimilar developers and manufacturers across North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa, including both pure-play biosimilar companies and diversified biopharmaceutical firms with biosimilar portfolios
Product Segmentation: Detailed mapping across monoclonal antibodies (anti-TNF, anti-HER2, anti-VEGF), recombinant growth factors (G-CSF, erythropoietin), recombinant hormones (insulin, growth hormone, follicle-stimulating hormone), and interferon categories
Revenue Analysis: Compilation of reported and modeled annual revenues specific to biosimilar product lines, incorporating disclosed tender awards, wholesale acquisition costs (WAC), and net pricing after rebates
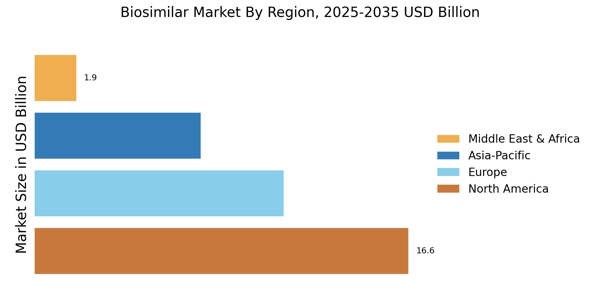
Market Coverage: Analysis encompassed manufacturers representing 75-80% of global biosimilar market share in 2024
Validation Approach: Extrapolation utilizing bottom-up methodology (target patient population × biosimilar penetration rate × average selling price by country/region) cross-validated with top-down manufacturer revenue aggregation and third-party pharmaceutical expenditure data to derive segment-specific valuations and growth projections
The methodology incorporated specific adjustments for regulatory exclusivity expiries, patent litigation settlements, interchangeability designations (US-specific), and biosimilar uptake curves observed in European reference markets versus emerging economies.