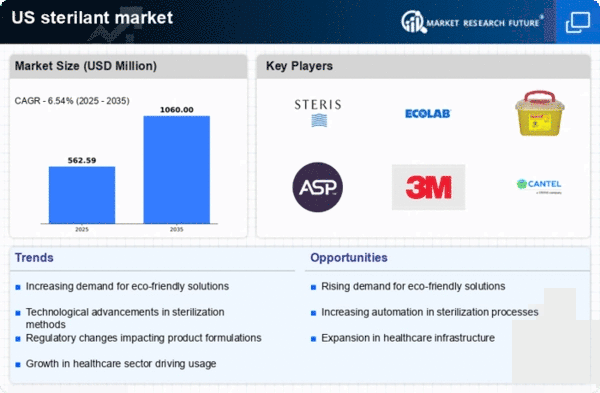
US Chemicals and Materials Market Segmentation
Chemicals and Materials By Application (USD Million, 2025-2035)
- Medical
- Pharmaceutical
- Food Processing
- Laboratory
- Agriculture
Chemicals and Materials By End Use (USD Million, 2025-2035)
- Hospitals
- Clinics
- Pharmaceutical Companies
- Food and Beverage Industry
- Research Institutions
Chemicals and Materials By Type of Sterilant (USD Million, 2025-2035)
- Ethylene Oxide
- Hydrogen Peroxide
- Formaldehyde
- Peracetic Acid
- Gamma Radiation
Chemicals and Materials By Formulation (USD Million, 2025-2035)
- Liquid
- Gas
- Solid
- Aerosol
Chemicals and Materials By Regulatory Compliance (USD Million, 2025-2035)
- FDA Approved
- EPA Registered
- ISO Certified
- CE Marked