ID: MRFR/MED/15162-HCR
100 Pages
Satyendra Maurya
Last Updated: April 06, 2026
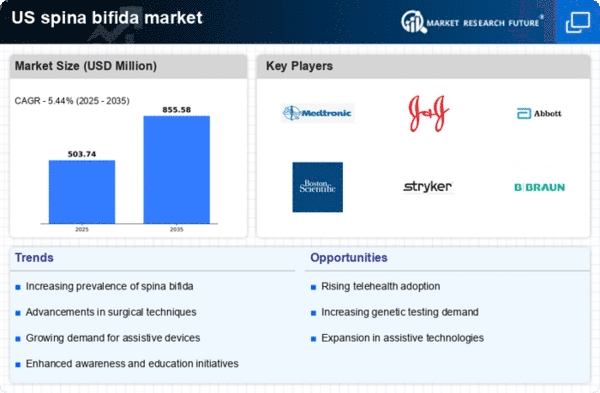
US Spina Bifida Market Research Report By Type (occulta, closed neural tube defects, meningocele, myelomeningocele), By Diagnosis (blood tests, ultrasound, amniocentesis, others, maternal serum alpha-fetoprotein (MSAFP) test, test to confirm high AFP levels, other blood tests), By Treatment (medical procedure, surgery, oral medications, others, urinary catheterization, urinary diversion, enema, spine surgery, fetal endoscopic surgery, spinal fusion, suprapubic cystostomy, caesarean section, antibiotics) and By End User (hospitals, clinics, diagnostic centers, drug stores, pharmacies, others) - Growth & Industry Forecast 2025 To 2035