US Motor Control Centers Market
ID: MRFR/EnP/14404-HCR
100 Pages
Snehal Singh
Last Updated: April 06, 2026
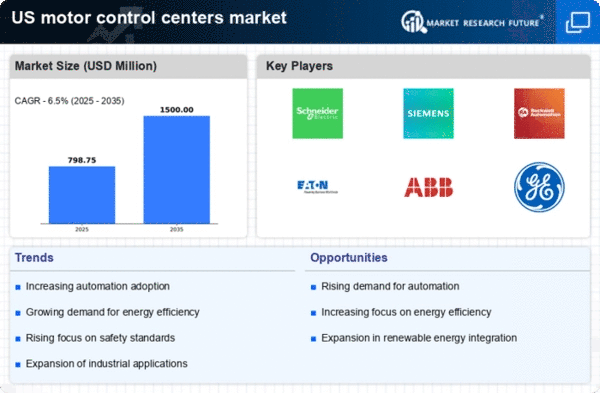
US Motor Control Centers Market Research Report By Type (Conventional MCC, Intelligent MCC), By Voltage (Low voltage, Medium voltage), By Component (Busbars, Circuit Breakers, Fuses, Overload Relays, Variable Speed Drives, Soft Starters, Others), By End User (Industrial, Commercial, Oil & Gas, Metal & Mining, Utilities, Chemical and Petrochemicals, Cement, Manufacturing Food & Beverage, others) and By Standard Segment (NEMA, IEC, Others) - Forecast to 2035