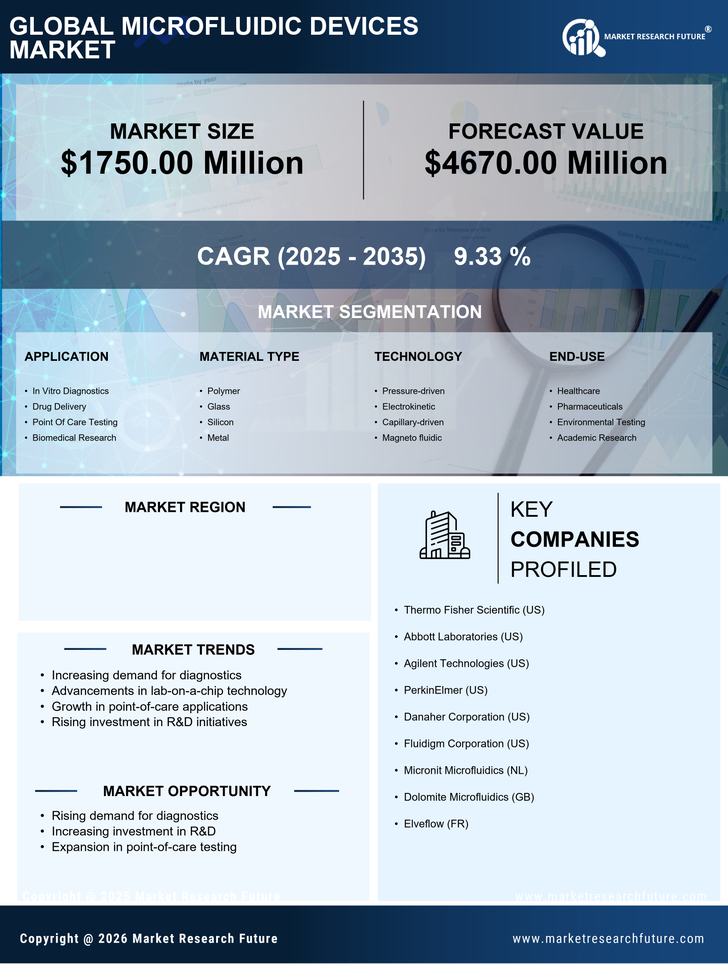
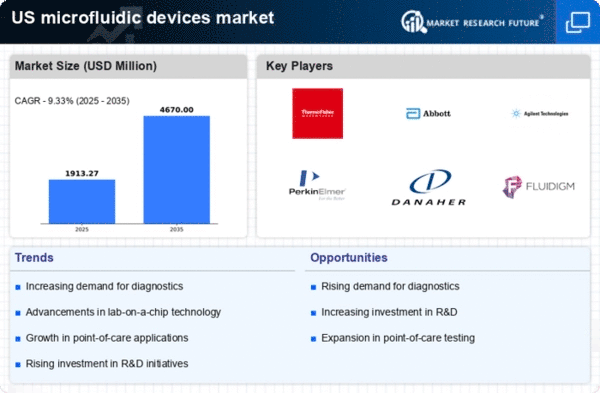
US Medical Device Market Segmentation
Medical Device By Application (USD Million, 2025-2035)
- In Vitro Diagnostics
- Drug Delivery
- Point Of Care Testing
- Biomedical Research
Medical Device By Material Type (USD Million, 2025-2035)
- Polymer
- Glass
- Silicon
- Metal
Medical Device By Technology (USD Million, 2025-2035)
- Pressure-driven
- Electrokinetic
- Capillary-driven
- Magneto fluidic
Medical Device By End-use (USD Million, 2025-2035)
- Healthcare
- Pharmaceuticals
- Environmental Testing
- Academic Research