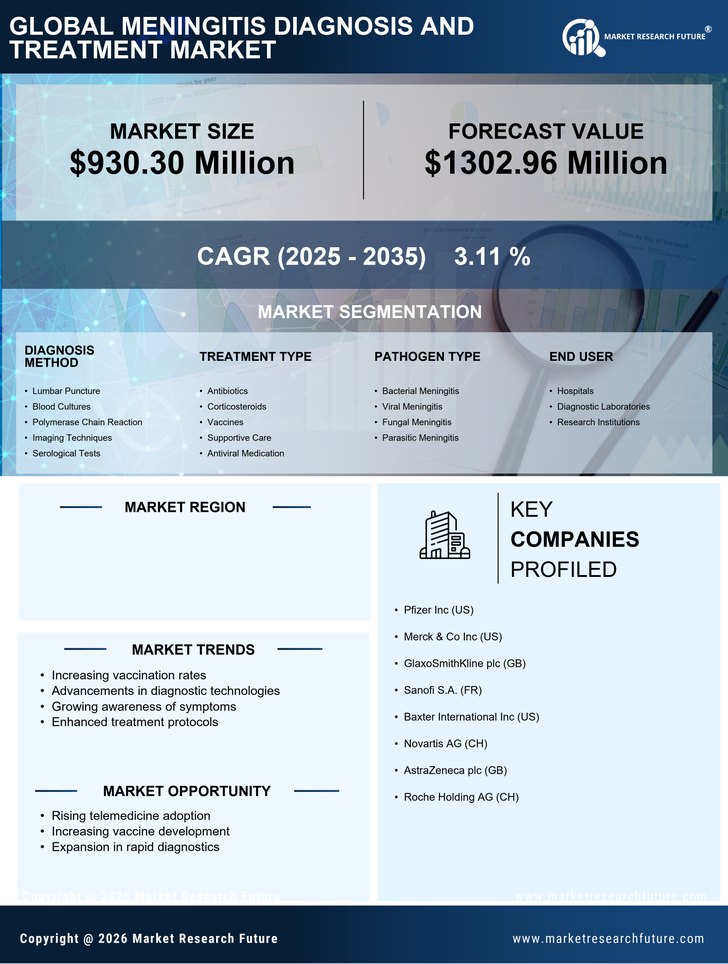
US Medical Device Market Segmentation
Medical Device By Diagnosis Method (USD Million, 2025-2035)
- Lumbar Puncture
- Blood Cultures
- Polymerase Chain Reaction
- Imaging Techniques
- Serological Tests
Medical Device By Treatment Type (USD Million, 2025-2035)
- Antibiotics
- Corticosteroids
- Vaccines
- Supportive Care
- Antiviral Medication
Medical Device By Pathogen Type (USD Million, 2025-2035)
- Bacterial Meningitis
- Viral Meningitis
- Fungal Meningitis
- Parasitic Meningitis
Medical Device By End User (USD Million, 2025-2035)
- Hospitals
- Diagnostic Laboratories
- Research Institutions