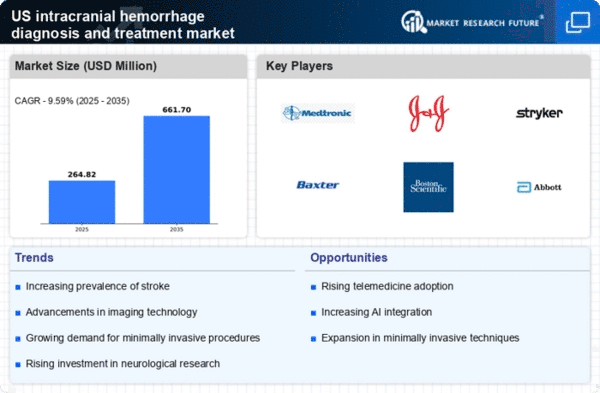
US Pharmaceutical Market Segmentation
Pharmaceutical By Type (USD Million, 2025-2035)
- Intracerebral Hemorrhage
- Subarachnoid Bleeding
- Epidural Hematoma
- Subdural Blood Hematoma
- Postpartum hemorrhage
- Pulmonary hemorrhage
Pharmaceutical By Devices (USD Million, 2025-2035)
- Invasive
- Non-invasive
Pharmaceutical By Diagnosis and Treatment (USD Million, 2025-2035)
- Diagnosis
- Treatment