ID: MRFR/HC/16521-CR
100 Pages
Satyendra Maurya
Last Updated: January 13, 2026
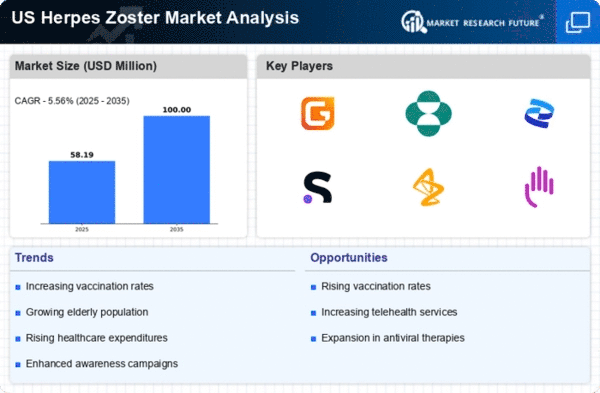
US Herpes Zoster Market Research Report By End User (Hospitals & Clinics, Diagnostic Centers, Research & Academic Institutes, Home Care Settings, Others) By Diagnosis (Laboratory Testing, Immunofluorescent Microscopy, Real-Time Polymerase Chain Reaction (PCR), Serological Testing, Others) By Route of Administration (Oral, Topical, Others) By Treatment and Prevention (Antiviral Medications, Narcotic Medications, Anti-Inflammatory Medications, Antihistamines, Anticonvulsants, Capsaicin, Numbing Creams, Gels, or Patches, Vaccines, Others) - Growth & Industry Forecast 2025 To 2035